Articles
- Page Path
- HOME > Restor Dent Endod > Volume 27(6); 2002 > Article
- Original Article A study of ionic dissociation on various calcium hydroxide pastes using molecular sieving model
- Kyoung-Sun Lee, D.D.S., M.S.D., Seung-Jong Lee, D.D.S., M.S.D., M.S., Ph D.
-
2002;27(6):-643.
DOI: https://doi.org/10.5395/JKACD.2002.27.6.632
Published online: November 30, 2002
Department of Dentistry, The graduate School, Yonsei University, Korea.
Copyright © 2002 Korean Academy of Conservative Dentistry
- 1,546 Views
- 7 Download
- 1 Crossref
Abstract
-
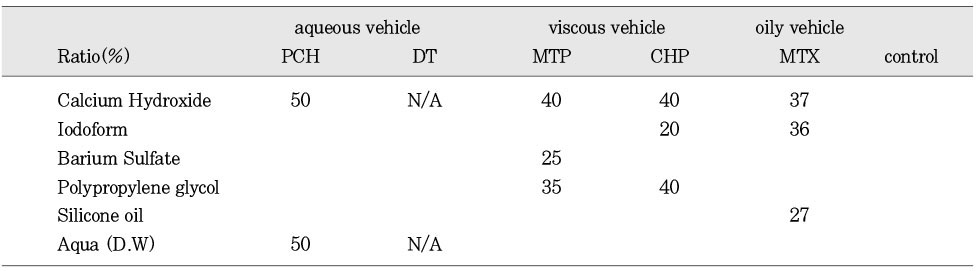
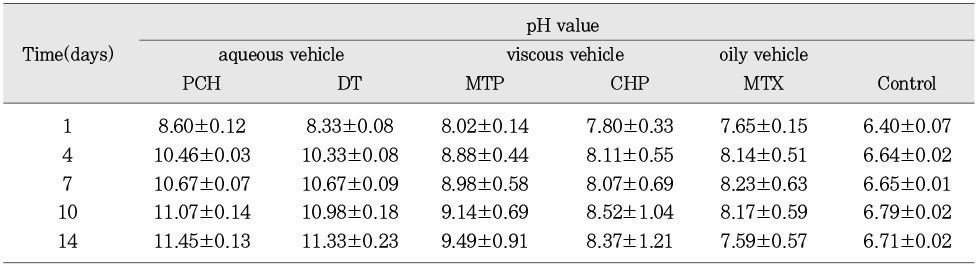
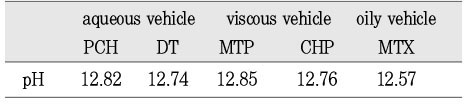
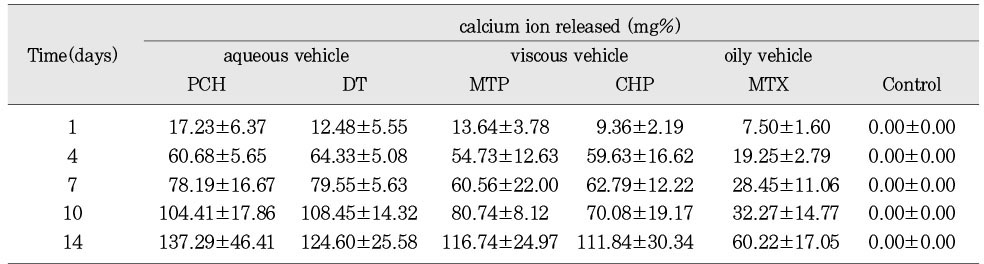
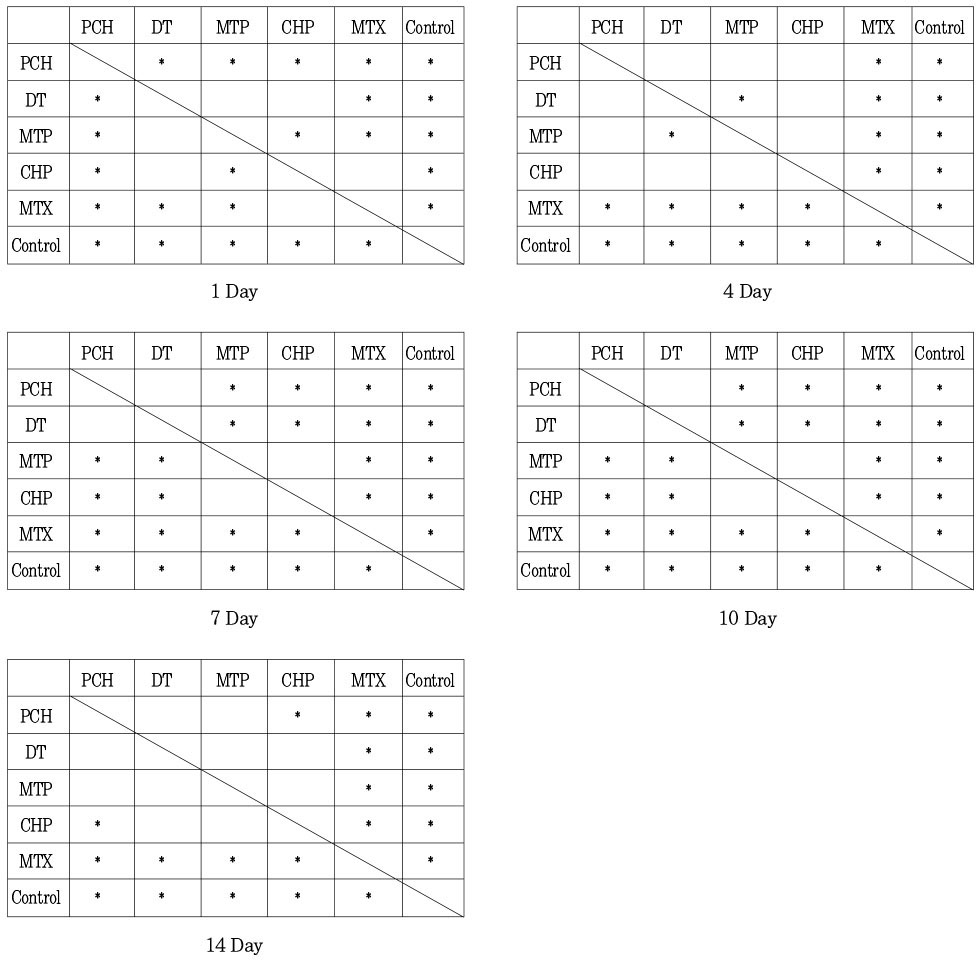
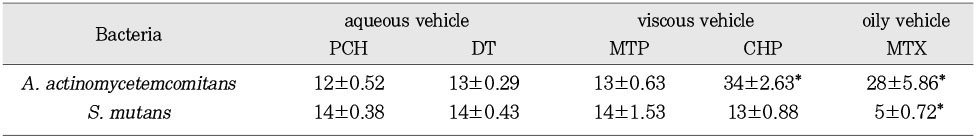
The purpose of this study was two-fold. First was to evaluate whether the molecular sieving model was appropriate for ionic dissociation experiment. Second was to compare the dissociation of calcium and hydroxyl ions from five types of calcium hydroxide pastes (Pure calcium hydroxide paste, DT temporary dressing®, Metapaste®, Chidopex®, Metapex®) in three vehicles (aqueous, viscous and oily) and the antibacterial effect.Each calcium hydroxide pastes was placed into 0.65ml tube with cap and then 15% polyacrylamide gel was placed onto calcium hydroxide pastes. After the gel was hardened, the tubes were filled with tridistilled water (pH 7.14) and closed with cap. The tubes were stored in 37℃, 100% incubator. The pH reading and the concentration of calcium ions were taken at 1, 4, 7, 10, and 14 days. The brain heart infusion agar plates with S. mutans and A. actinomycetemcomitans were used for antibacterial activity test. Middle of agar plate was filled with the calcium hydroxide pastes. The plates were incubated at 37℃ and observations were made to detect the zones of inhibition. These data were evaluated statistically by use of the analysis of variance and duncan test.The results were as follows.1. In fresh mixing state, the pH of five types of calcium hydroxide pastes were measured between 12.5 and 12.8.2. The pH was increased in all five types of calcium hydroxide pastes compared with control group. In 14 days, Pure calcium hydroxide paste (11.45) and DT temporary dressing® (11.33) showed highest pH, followed by Metapaste® (9.49), Chidopex® (8.37) and Metapex® (7.59).3. Calcium was higher in all five types of calcium hydroxide pastes compared with control group. In 14 days, Pure calcium hydroxide paste (137.29 mg%) and DT temporary dressing® (124.6 mg%) showed highest value, followed by Metapaste® (116.74 mg%), Chidopex® (111.84 mg%) and Metapex® (60.22 mg%).4. The zones of bacterial inhibition were seen around all five types of calcium hydroxide pastes. Chidopex® and Metapex® groups which include iodoform were observed significantly larger zone of inhibition in A. actinomycetemcomitans compared with the other calcium hydroxide groups (p<0.05). However, Metapex® showed the least antibacterial effect on S. mutans compared with other groups (p<0.05).The molecular sieving model was found to be acceptable in dissociation experiment of hydroxyl and calcium ions when compared with the previous tooth model study. But this model was not appropriate for the antibacterial test.
- 1. Binnie WH, Rowe AHR. A histological study of the periapical tissues of incompletely formed pulpless teeth filled with calcium hydroxide. J Dent Res. 1973;52: 1110-1116.ArticlePubMedPDF
- 2. Erdogan G. The treatment of nonvital immature teeth with calcium hydroxide-sterile water paste. Quintessence Int. 1997;28: 681-686.PubMed
- 3. Mitchell DF, Shankwalker GB. Osteogenic potential of calcium hydroxide and other materials in soft tissues and bone wounds. J Dent Res. 1958;37: 1157-1163.ArticlePubMedPDF
- 4. Hasselgren G, Olsson B, Cvek M. Effects of calcium hydroxide and sodium hypochlorites in the dissolution of necrotic porcine muscle tissue. J Endod. 1988;14: 125-127.PubMed
- 5. Türkün M, Cengiz T. The effects of sodium hypochlorite and calcium hydroxide on tissue dissolution and root canal cleanliness. Int Endod J. 1997;30: 335-342.ArticlePubMed
- 6. Morgan RW, Carnes DL, Montgomery S. The solvent effects of calcium hydroxide irrigating solution on bovine pulp tissue. J Endod. 1991;17: 165-168.ArticlePubMed
- 7. Calişkan MK, Şen BH. Endodontic treatment of teeth with apical periodontitis using calcium hydroxide calcium hydroxide: a long-term study. Endod Dent Traumatol. 1996;12: 215-221.PubMed
- 8. Lengheden A, Blomlöf L, Lindskog S. Effect of immediate calcium hydroxide treatment and permanent root-filling on periodontal healing in contaminated replanted teeth. Scand J Dent Res. 1990;99: 139-146.Article
- 9. Chong BS, Pittford TR. The role of intracanal medication in root canal treatment. Int Endod J. 1992;25: 97-106.ArticlePubMed
- 10. Walton RE. Intracanal medications. Dent Clin North Am. 1984;28: 783-796.PubMed
- 11. Montgomery S. External cervical resorption after bleaching a pulpless tooth. Oral Surg Oral Med Oral Pathol. 1984;57: 203-206.ArticlePubMed
- 12. Gimlin DR, Schindler WG. The management of postbleaching cervical resorption. J Endod. 1990;16: 292-297.ArticlePubMed
- 13. Tronstad L, Andreasen JO, Kristerson L, Riis I. pH changes in dental tissues after root canal filling with calcium hydroxide. J Endod. 1981;7: 17-21.ArticlePubMed
- 14. Massarstrom LE. Effect of calcium hydroxide treatment on periodontal repair and root resorption. Endod Dent Traumatol. 1986;2: 184-189.ArticlePubMed
- 15. Fuss Z, Szajkis S, Tagger M. Tubular permeability to calcium hydroxide and to bleaching agents. J Endod. 1989;15: 362-364.ArticlePubMed
- 16. Wang JD, Hume WR. Diffusion of hydrogen and hydroxyl ions from various sources through dentine. Int Endod J. 1988;21: 17-26.PubMed
- 17. Javelet J, Torabinejad M, Bakland LK. Comparison of two pH levels for the induction of apical barriers in immature teeth of monkeys. J Endod. 1985;11: 375-378.ArticlePubMed
- 18. Kawakami T, Nakamura C, Hasegawa H, Eda S. Fate of 45Ca-labeled calcium hydroxide in a root canal filling paste embedded in rat subcutaneous tissues. J Endod. 1987;13: 220-223.ArticlePubMed
- 19. Foster KH, Kulid JC, Weller RN. Effect of smear layer removal on the diffusion of calcium hydroxide through radicular dentin. J Endod. 1993;19: 136-140.ArticlePubMed
- 20. Kwon BG. The influence of smear layer upon dentin diffusion of calcium hydroxide in bovine tooth in vitro. J Korean Acad Conserv Dent. 1997;22: 111-131.
- 21. Nerwich A, Figdor D, Endo D, Messer HH. pH changes in root dentin over a 4-week period following root canal dressing with calcium hydroxide. J Endod. 1993;19: 302-306.ArticlePubMed
- 22. Stuart KG, Miller CH, Brown CE Jr, Newton CW. The comparative antimicrobial effect of calcium hydroxide. Oral Surg Oral Med Oral Pathol. 1991;72: 101-104.ArticlePubMed
- 23. Thomas PA, Bhat KS, Kotian KM. Antibacterial properties of dilute formocresol and eugenol and propylene glycol. Oral Surg Oral Med Oral Pathol. 1980;49: 166-170.ArticlePubMed
- 24. Stevens RH, Grossman LI. Evaluation of the antimicrobial potential of calcium hydroxide as an intracanal medicament. J Endod. 1983;9: 372-374.ArticlePubMed
- 25. Alaçam T, Görgül G, Ömürlü H. Evaluation of diagnostic radiopaque contrast materials used with calcium hydroxide. J Endod. 1990;16: 365-368.ArticlePubMed
- 26. Smith GN, Woods S. Organic iodine: A substitute for BaSO4 in apexification procedures. J Endod. 1983;9: 153-155.ArticlePubMed
- 27. Rivera EM, Williams K. Placement of calcium hydroxide in simulated canals:comparison of glycerin versus water. J Endod. 1994;20: 445-448.ArticlePubMed
- 28. Barbosa CAM, Gonc¸alves RB, Siqueira JF Jr, De Uzeda M. Evaluation of the antibacterial activities of calcium hydroxide, chlorhexidine and camphorated paramonochlorophenol as intracanal medicament. A clinical and laboratory study. J Endod. 1997;23: 297-300.ArticlePubMed
- 29. Fava LRG, Saunders WP. Calcium hydroxide pastes:classification and clinical indications. Int Endod J. 1999;32: 257-282.ArticlePubMed
- 30. Anthony DR, Gordon TM, del Rio CE. The effect of three vehicles on the pH of calcium hydroxide. Oral Surg Oral Med Oral Pathol. 1982;54: 560-565.ArticlePubMed
- 31. Simon ST, Bhat KS, Francis R. Effect of four vehicles on the pH of calcium hydroxide and the release of calcium ion. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;80: 459-464.ArticlePubMed
- 32. Gordon TM, Ranly DM, Boyan BD. The effects of calcium hydroxide on bovine pulp tissue. J Endod. 1985;11: 156-160.PubMed
- 33. Lengheden A, Blomlöf L, Lindskog S. Effect of delayed calcium hydroxide treatment on periodontal healing in contaminated replanted teeth. Scand J Dent Res. 1991;99: 147-153.ArticlePubMed
- 34. Lengheden A. Influence of pH and calcium on growth and attachment of human fibroblasts in vitro. Scand J Dent Res. 1994;102: 130-136.ArticlePubMed
- 35. Lengheden A, Jansson L. pH effects on experimental wound healing of human fibroblasts in vitro. Eur J Oral Sci. 1995;103: 148-155.ArticlePubMed
- 36. Carrigan PJ, Morse DR, Furst ML, Sinai IH. A scanning electron microscopic evaluation of human dentinal tubules according to age and location. J Endod. 1984;10: 359-363.ArticlePubMed
- 37. Tamburic SD, Vuleta GM, Ognjanovic JM. In vitro release of calcium and hydroxyl ions from two types of calcium hydroxide preparation. Int Endod J. 1993;26: 125-130.ArticlePubMed
- 38. Larsen MJ, Hrsted-Bindslev P. A laboratory study evaluating the release of hydroxyl ions from various calcium hydroxide products in narrow root canallike tubes. Int Endod J. 2000;33: 238-242.ArticlePubMed
- 39. Beltes PG, Pissiotis E, Koulaouzidou E, Kortsaris AH. In vitro release of hydroxyl ions from six types of calcium hydroxide nonsetting pastes. J Endod. 1997;23: 413-415.ArticlePubMed
- 40. Fuss Z, Rafaeloff R, Tagger M, Szajkis S. Intracanal pH changes of calcium hydroxide pastes exposed to carbon dioxide in vitro. J Endod. 1996;22: 362-364.ArticlePubMed
- 41. Byström A, et al. The antibacterial effect of camphorated paramonochlorophenol, camphorated phenol and calcium hydroxide in the treatment of infected root canals. Endod Dent Traumatol. 1985;1: 170-175.ArticlePubMed
- 42. Sjgren U, Fogdor D, Spangberg L, Sundqvist G. The antimicrobial effect of calcium hydroxide as a short-term intracanal dressing. Int Endod J. 1991;24: 119-125.ArticlePubMed
- 43. Heithersay GS. Calcium hydroxide in the treatment of pulpless teeth with associated pathology. J Br Endod Soc. 1975;8: 74-93.ArticlePubMed
- 44. Siqueira JF, de Uzeda M. Disinfection by calcium hydroxide pastes of dentinal tubules infected with two obligate and one facultative anaerobic bacteria. J Endod. 1996;22: 674-676.ArticlePubMed
- 45. Rehman K, Saunders WP, Foye RH, Sharkey SW. Calcium ion diffusion from calcium hydroxide-containing materials in endodontically-treated teeth : An in vitro study. Int Endod J. 1996;29: 271-279.ArticlePubMed
- 46. Macdonald JB, Hare GC, Wood AWS. The bacteriologic status of the pulp chambers in intact teeth found to be nonvital following trauma. Oral Surg Oral Med Oral Pathol. 1957;10: 318-322.ArticlePubMed
- 47. Foreman PC, Barnes IE. A review of calcium hydroxide. Int Endod J. 1990;23: 283-297.ArticlePubMed
REFERENCES
Tables & Figures
REFERENCES
Citations

- A Comparison of the irrigation systems in calcium hydroxide removal
Jae-Seung Eun, Se-Hee Park, Kyung-Mo Cho, Jin-Woo Kim
Journal of Korean Academy of Conservative Dentistry.2009; 34(6): 508. CrossRef

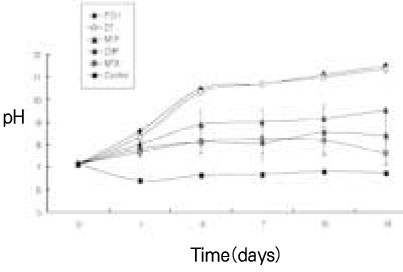

Fig. 1
Fig. 2
Fig. 3
The solutions for preparing 15% polyacrylamide gel (10ml)
The components of the calcium hydroxide paste(%)
*N/A : Calcium hydroxide and distilled water are mixed, pH 12.6
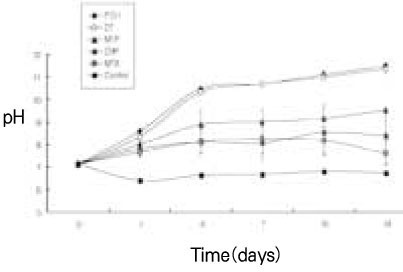
pH values in released solution of calcium hydroxide (mean ± S.D.)
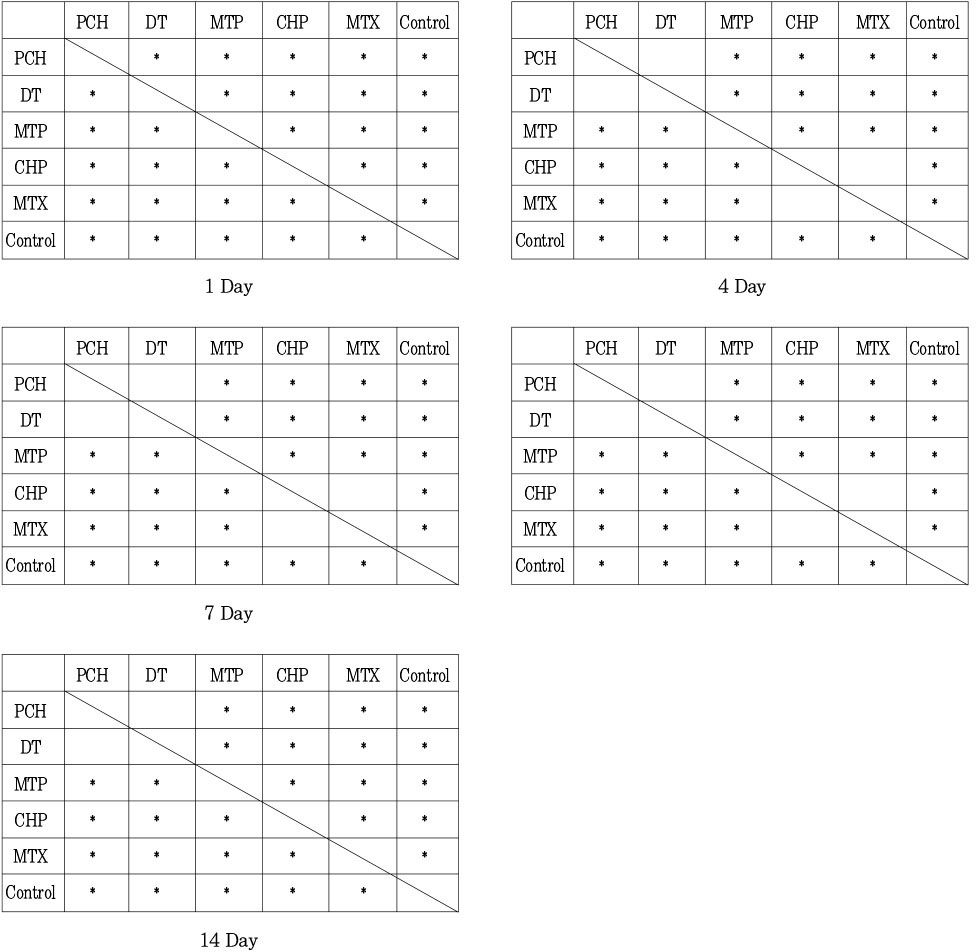
Statistical differences between test groups in pH by duncan test Statistically significant differences (p<0.05) are shown by *.
pH values of calcium hydroxide pastes (fresh mixing state)
Concentration of calcium ion in released solution of calcium hydroxide pastes. (mean ± S.D.)
Statistical differences between test groups in concentration of claciumions. Statistically significant differences (p<0.05) are shown by *.
Inhibitory effect of calcium hydroxide pastes on proliferation of S. mutans and A. actinomycetemcomitans.(mean ± S.E.)
*: Statistically significant difference, p<0.05 by Mann-Whitney U-test
※The data is expressed as diameter of inhibition zone (mm).
*N/A : Calcium hydroxide and distilled water are mixed, pH 12.6
*: Statistically significant difference, p<0.05 by Mann-Whitney U-test ※The data is expressed as diameter of inhibition zone (mm).

 KACD
KACD



 ePub Link
ePub Link Cite
Cite