Abstract
-
Objectives
This in vitro study aimed to assess the fracture resistance of simulated stages of root maturation following regenerative endodontic treatment using a cyclic loading method.
-
Methods
Ninety extracted maxillary central incisors were randomly allocated into three experimental groups representing different stages of root development, following revitalization: Group A for completely immature teeth immediately after treatment; Group B for teeth with apical closure, and Group C for teeth with apical closure and wall thickening. Two control groups were also included: Group D for intact teeth and Group E for simulated immature teeth without the bioceramic material. Following simulation of immature apices and treatment with a bioceramic material, all specimens were subjected to cyclic loading using a step-stress fatigue protocol until failure. The number of cycles to fracture and the peak load were recorded and statistically analyzed.
-
Results
Statistically significant differences in loading forces were observed between the negative control group (Group D) and Groups A, B, and E (p < 0.05). However, no statistically significant differences were detected among the experimental groups. These results indicate that apical closure and dentinal wall thickening alone did not substantially improve mechanical reinforcement under cyclic loading conditions.
-
Conclusions
Although intact teeth exhibited superior mechanical performance, apical closure and wall thickening alone were insufficient to enhance reinforcement under cyclic loading.
-
Keywords: Tooth fractures; Fracture strength; Regenerative endodontics; In vitro techniques
INTRODUCTION
Pulpal necrosis in immature teeth, often caused by trauma, caries, or developmental disorders, presents significant clinical challenges. Incomplete root formation results in thin dentinal walls and open apices, making these teeth more susceptible to fractures, particularly in the cervical region [
1]. Finite element analyses have shown that the highest stress concentrations occur in the coronal portion of the root when subjected to masticatory or traumatic forces [
2]. Consequently, fractures frequently develop at the level of the crestal bone, rendering restoration impossible and compromising the long-term prognosis of the tooth [
3].
For immature non-vital teeth, current treatment options include regenerative endodontic procedures (REPs) and apexification, typically achieved using a mineral trioxide aggregate (MTA) plug or calcium hydroxide [Ca(OH)₂]-induced apical barrier formation [
4]. However, the use of MTA apexification has declined due to its high cost, complex handling characteristics, retreatability challenges, and risk of overfilling [
5]. Clinical studies comparing the survival and success rates of REPs and apexification have shown similar overall outcomes [
6].
Nevertheless, neither Ca(OH)₂ nor MTA apexification supports continued root development [
7]. In contrast, REPs aim to restore pulp vitality and promote root maturation by stimulating increases in root length, dentinal wall thickness, and apical closure. Furthermore, REPs offer an advantage by allowing for potential retreatment if the initial procedure fails [
8,
9]. Despite these benefits, the long-term effects of REPs on the structural integrity of treated teeth remain uncertain. In particular, the relationship between the degree of root maturation and the fracture resistance of healing or healed immature teeth remains unclear.
Therefore, this study aims to evaluate the fracture resistance of revascularized teeth at three simulated stages of root development: completely immature, with apical closure, and with apical closure and dentine wall thickening, under cyclic loading conditions. The null hypothesis tested was that apical closure, with or without dentinal wall thickening, does not significantly influence the fracture resistance of the tooth.
METHODS
Study design
The study was conducted in compliance with the ethical standards of the Aristotle University of Thessaloniki Research Committee (approval number: 222/13-03-2024) and the 1964 Helsinki Declaration and its later amendments. The manuscript was prepared following the Preferred Reporting Items for Laboratory Studies in Endodontology 2021 guidelines [
10]. All teeth were collected with informed consent from patients undergoing extraction for reasons unrelated to this study, ensuring patient confidentiality throughout the research process.
A sample size calculation was performed using G*Power software (ver. 3.1.9.7, Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany), as indicated by previous published studies comparing fracture resistance with various regenerative endodontic protocols [
11,
12]. Accordingly, a sample size of 15 teeth per experimental group and 10 per control group was determined to achieve 80% power at a significance level of 0.05. The number of samples increased by five in each group (×5 groups) to account for potential laboratory processing errors.
Ninety freshly extracted intact maxillary central incisors were collected for the study. Calculus and soft tissue deposits were removed with an ultrasonic device (Woodpecker Ultrasonic Scaler; Guilin Woodpecker, Guilin, China). The teeth were sterilized in an autoclave (Lisa; W&h, Bürmoos, Austria) for 40 minutes at 134°C under 20 psi, then stored in distilled water at 4°C [
13]. Prior to experimentation, the teeth were carefully examined under magnification (SmartOPTIC; Seliga Microscopes, Lodz, Poland) to ensure they were free from resorption, caries, cracks, or deformities. Teeth from the same individual were excluded.
To ensure specimen homogeneity prior to group allocation, all teeth were measured using a digital caliper (Mitutoyo 150 mm Digital Caliper; Mitutoyo, Hampshire, UK) in three dimensions—buccolingual, mesiodistal, and corono-apical (height)—to establish mean values for the sample. Specimens deviating more than 20% from the mean in any dimension or exhibiting canal curvature greater than 20° were excluded to minimize variation in root size and dentinal wall morphology that could influence stress distribution during fracture testing. The final sample demonstrated consistent morphology, with mean mesiodistal dimensions of 6.5 ± 0.5 mm and buccolingual dimensions of 6.0 ± 0.5 mm. Mesiodistal and buccolingual measurements were recorded at the cementoenamel junction (CEJ), whereas the corono-apical dimension corresponded to the straight line connecting the incisal edge to the apical tip. Each measurement was performed five times; the highest and lowest values were discarded, and the remaining three were averaged to ensure accuracy.
All specimens were radiographed in buccolingual and mesiodistal projections using a digital sensor (Planmeca ProSensor; Planmeca, Helsinki, Finland) at three stages: before instrumentation to verify root length, assess canal morphology, and screen for differences in dentinal wall thickness or pre-existing defects; after bioceramic placement to verify material thickness and homogeneity; and after fracture testing to confirm failure patterns. This imaging-based verification further minimized anatomical variability that could affect stress distribution and fracture resistance. The combination of morphometric caliper measurements and radiographic evaluation ensured consistent root dimensions and dentinal wall thickness across specimens, thereby reducing anatomical variability that could influence fracture resistance outcomes. After confirming anatomical comparability, the specimens were randomly assigned to five groups—three experimental groups (n = 20 each) and two control groups (n = 15 each)—using computer-generated randomization software (QuickCalcs, GraphPad Software, San Diego, CA, USA).
Simulation of immature teeth
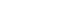
Experimental groups were instrumented in order to represent three different stages of root development: completely immature teeth (Group A), teeth with apical closure (Group B), and teeth with apical closure and wall thickening (Group C). Intact teeth served as negative controls (Group D), while teeth with simulated immature roots without material placement served as positive controls (Group E) (
Figure 1). The completely immature groups were classified as stage 3 according to Cvek’s classification [
14].
The apices of the teeth in the positive control and Group A (completely immature) were cut by a low-speed diamond disc (Superflex HP 405; Edenta AG, Pfungen, Austria) such that the remaining root length was 12 mm [
15,
16]. Immature teeth were simulated according to the study of Elnaghy and Elsaka [
16]. Briefly, specimens were enlarged from the coronal to apical direction using long-neck round burs at high-speed rotation with continuous air-water spray. Peeso reamers (Dentsply Maillefer, Ballaigues, Switzerland) of size 1–6 were inserted into varying root canal lengths depending on the experimental group: Groups A and E were instrumented until size 6 Peeso to root length; for Group B, 3 mm coronal to the apex; and for Group C, 3 mm apical to the CEJ. Instrumentation was performed by a single experienced operator using Peeso reamers with fixed, standardized diameters to ensure reproducible enlargement patterns across all specimens [
12,
15]. Each canal was prepared following a predefined depth-specific protocol to produce uniform tapering and to maintain consistent dentinal wall thickness within and between groups. The use of dimensionally calibrated reamers and a single operator minimized procedural variability and maximized specimen homogeneity, ensuring that differences in dentinal wall size did not confound the experimental outcomes.
The treatment procedures were conducted in accordance with the protocol recommended by the European Society of Endodontology (ESE) [
17]. Following access cavity preparation, the root canals were rinsed with 1.5% sodium hypochlorite (20 mL, 5 minutes), using a side-vented needle, followed by 5 mL sterile saline and 20 mL 17% ethylenediaminetetraacetic acid (EDTA) (CanalPro EDTA 17%; COLTENE Group, Cuyahoga Falls, OH, USA). The canals were dried with paper points (Diadent Paper Points; Diadent Europe B.V., Almere, The Netherlands) and filled with calcium hydroxide as an intracanal medicament following the protocol of Jamshidi
et al. [
15]. Access cavities were provisionally restored using a temporary filling material (Cavit; 3M ESPE, St Paul, MN, USA).
The teeth were then incubated at 37°C and 100% humidity for 3 weeks. After the incubation period, the calcium hydroxide paste was removed using 1.5% sodium hypochlorite. The root canals were then irrigated with 17% EDTA and distilled water, then dried with paper points. A foam was placed in the root canal up to the working length to simulate the blood clot, standardize the coronal barrier thickness, and facilitate material condensation, following the methodology described by Gunal
et al. [
12].
A 3-mm layer of the bioceramic material (Angelus Bio-C Repair; Angelus, Londrina, Brazil) was applied according to the manufacturer’s instructions in all three experimental groups below the CEJ. Teeth were temporally restored with Cavit. To simulate clinical conditions, all specimens were stored in 100% humidity at 37°C until fracture testing. After 12 hours of incubation to allow the bioceramic putty to set, access cavities were restored with glass ionomer cement (Ionoseal; Voco, Cuxhaven, Germany) and light-cured flowable composite (Spectrum; Dentsply Sirona, Constanz, Germany).
Fracture resistance testing
For periodontal ligament (PDL) simulation, teeth were coated with a latex liquid spacing agent (Erkoskin; Erkodent, Pfalzgrafenweiler, Germany) according to the method described by Mello
et al. [
11]. Each tooth was coated with four layers of Erkoskin, with 10 minutes of drying between layers. The specimens were then embedded perpendicularly in self-curing acrylic resin (BMS Light-curing orthodontic resin; BMS Dental, Capannoli, Italy), leaving a 2-mm gap between the top of the acrylic and the CEJ to mimic the bone-tooth relationship. For fracture testing, a custom jig was prepared to fix the acrylic cubes at a 45° angle [
18]. Samples were then subjected to cyclic loading using a 2-mm spherical stainless-steel tip mounted on a pneumatic cylinder (SDA; Airblock, Thessaloniki, Greece), controlled by a precision pressure regulator (Regtronic; Metal Work, Consecio, Italy). The pneumatic loading system was operated via a single-board computer running custom Python-based control software and was calibrated using a Universal Testing Machine (Testometric, Rochdale, UK). The pneumatic loading system was calibrated at regular intervals throughout the experimental procedure. Calibration checks were performed after every 10 specimens, and no deviations beyond acceptable tolerance limits were detected. This maintained consistent and reliable loading throughout the experiment. To ensure constant moisture conditions during testing, all samples were covered with a wet gauze throughout the procedure [
19].
Fracture resistance was evaluated using the step-stress method described by Lin
et al. [
19] (
Supplementary Table 1), in which cyclic loads were progressively increased until specimen failure. The loading cycles followed a sinusoidal waveform at 1 Hz, with the minimum load set to 0. The test commenced with a load amplitude of 200 N, followed by 300 N, each applied for 1,000 cycles. Subsequently, the load amplitude was increased to 400 N and further raised in 50 N increments up to 850 N, with 3,000 cycles at each level. Specimens that withstood these cycles were loaded to 850 N until fracture occurred. A high-sensitivity sensor (G-Sensor; Creality, Shenzhen, China) was employed to detect subtle positional displacements of the cylinder tip indicative of fracture. Data on the number of cycles to failure and the final load at failure were recorded for analysis.
Descriptive statistics, including means and standard deviations, were calculated for fracture resistance values in each group. The normality of data distribution was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests. Since the data were not normally distributed, non-parametric tests were applied. Differences among the five experimental groups were analyzed using the Kruskal-Wallis test, followed by Bonferroni-adjusted Dunn’s post hoc comparisons for pairwise analysis. A p-value of <0.05 was considered statistically significant. Statistical analyses were performed using IBM SPSS ver. 28.0 (IBM Corp., Armonk, NY, USA).
RESULTS
Normality tests (Shapiro-Wilk and Kolmogorov-Smirnov) revealed that the data for cyclic load, fracture resistance, and number of cycles were not normally distributed (p < 0.05). The Kruskal–Wallis test followed by Bonferroni-adjusted Dunn’s post hoc comparisons was used to evaluate differences among the five experimental groups.
Fracture resistance
Descriptive statistics for mean load (N), fracture resistance (MPa), and coefficient of variation (CV%) are presented in
Table 1. The Kruskal-Wallis test revealed significant differences among the groups (H(4) = 20.270,
p < 0.05).
Post hoc pairwise comparisons using Bonferroni-adjusted Dunn’s tests showed that the negative control (Group D) exhibited significantly higher fracture resistance than Groups A (
p < 0.05), B (
p < 0.05), and E (
p < 0.05). No statistically significant differences were detected among the experimental groups (A–C) or between Group C and the negative control (Group D).
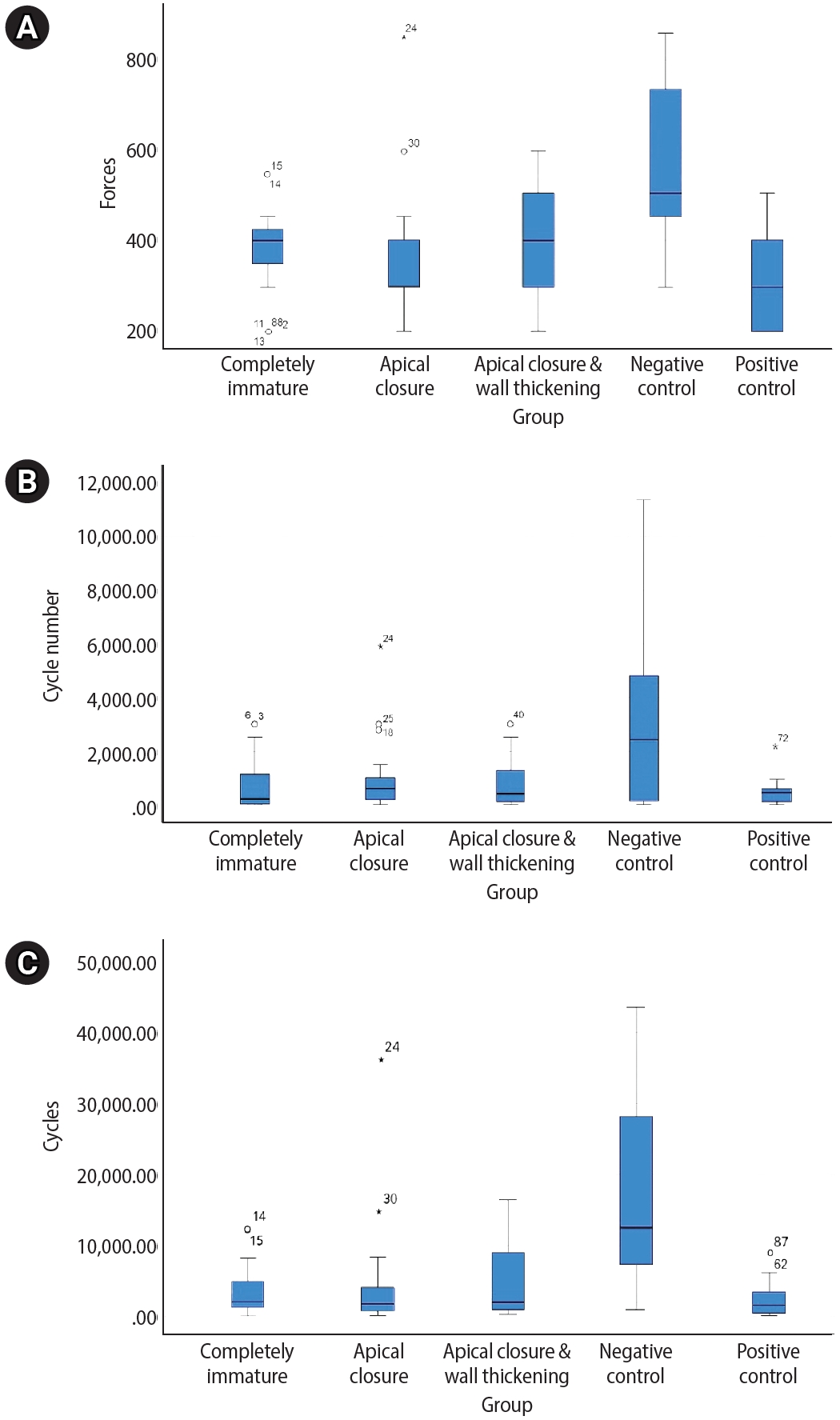
Group C displayed the highest mean fracture resistance among the experimental groups (5,019.05 ± 4,968.58 MPa) and was comparable to the negative control (15,513.33 ± 13,244.33 MPa). However, this observation should be interpreted cautiously, as differences among the experimental groups were not statistically significant (p > 0.05).
Load at failure
Mean load values per cycle also varied significantly among groups (H(4) = 20.332, p < 0.05). Group D (573.33 ± 186.96 N) had significantly higher load capacity compared with Groups A (377.5 ± 105.72 N, p < 0.05), B (362.5 ± 153.79 N, p < 0.05), and E (316.67 ± 109.65 N, p < 0.05). No significant differences were found between Groups A, B, and C.
Number of cycles to fracture
The number of cycles to failure did not differ significantly among groups (H(4) = 5.468, p > 0.05). The highest mean number of cycles was recorded for the negative control (2,972.33 ± 3,334.44), followed by Group C (821.50 ± 893.79). The lowest values were observed in the positive control (514.33 ± 549.95).
The distribution of mechanical parameters among the experimental groups is illustrated in
Figure 2. Boxplots show the variability in load at failure (
Figure 2A), fracture resistance (
Figure 2B), and number of cycles to fracture (
Figure 2C) across all five groups.
DISCUSSION
REPs offer a promising alternative to traditional root canal treatment, allowing for the regeneration of functional dental pulp [
20]. The effectiveness of REPs is assessed based on criteria established by the ESE and the American Association of Endodontists (AAE) [
17,
21]. The ESE defines success as clinical and radiographic healing, continued root development, a positive pulp sensibility response, absence of root resorption, and absence of crown discoloration [
17]. The AAE categorizes outcomes as primary (symptom resolution), secondary (root development), and tertiary (pulp sensibility) [
21]. To integrate these perspectives, the ESE criteria describe the observable clinical and radiographic manifestations of success, while the AAE framework contextualizes these changes within biologically driven stages of regeneration. Together, they provide a unified and comprehensive basis for evaluating treatment outcomes in REP cases.
Numerous studies report that REPs frequently result in apical closure, often accompanied by continued root development, including an increase in root length and wall thickness. However, variability exists: while some cases show complete maturation, others exhibit only apical closure without significant root elongation [
22,
23]. This variability highlights the need to understand which specific components of root development contribute meaningfully to mechanical reinforcement.
This in vitro study investigated the impact of different simulated stages of root development, specifically apical closure or a combination of apical closure and wall thickening, on the fracture resistance of simulated immature teeth treated with REP. The results demonstrated no statistically significant differences among the experimental groups, supporting the acceptance of the null hypothesis. Specifically, neither apical closure alone nor apical closure combined with wall thickening resulted in a statistically significant increase in fracture resistance compared with completely immature teeth. These findings help clarify the contribution of individual simulated growth elements to fracture resistance under laboratory conditions, an area that has remained ambiguous in previous literature.
The fracture resistance of immature teeth subjected to REPs has been evaluated in a limited number of research studies [
12,
13,
15,
16,
18,
24]. Most studies have shown that treated teeth have lower fracture resistance compared to intact teeth, an observation that aligns with the findings of the present study. Previous research did not find significant differences in fracture resistance based on the stage of root development [
15], the material used [
24], and material thickness [
12] or the presence of material at the critical cervical area [
16].
However, simulated immature teeth showed advanced fracture resistance when fully obturated with MTA [
12]. This highlights that material-based reinforcement is possible under certain conditions. In the present study, no significant difference was observed between the positive control (Group E) and the completely immature group (Group A), indicating that bioceramic materials placed at the coronal part of the root canal, below the CEJ, may not effectively strengthen canal walls. Only the negative control group exhibited significantly higher values, underscoring the superior integrity of unaltered dentine.
Τhe use of a bioceramic material at the cervical root area is considered essential for the REP protocol. Recently, there has been a push to develop improved biomaterials. Angelus Bio-C Repair, introduced in 2019, is a premixed paste of bioceramic repair material delivered via syringe. In this study, it was selected for its user-friendly handling characteristics and ease of clinical application. Regardless of the material used, a major limitation of REPs is the hindrance to posttreatment growth of pericervical dentine caused by the placement of bioceramic material. Pericervical dentine growth is crucial for safely transferring load from the occlusal table to the root [
25]. This fact is emphasized by the emerging long-term literature data that have identified cervical fractures as a notable cause of posttreatment failure [
26,
27].
Numerous studies have investigated the fracture resistance of teeth treated with regenerative protocols using static loading tests [
15,
28]. However, there is no consensus in the literature on the most accurate method for assessing fracture resistance. Static loading tests often overestimate fracture resistance [
29], whereas cyclic loading, which simulates oral mastication conditions, is limited by time and cost constraints. Accelerated life testing, such as step-stress testing, is commonly used to identify potential failure modes by subjecting specimens to extreme conditions. Step-stress testing involves applying a constant load for a period before increasing it until failure occurs [
30]. This approach has been successfully used to generate longitudinal cracks in endodontically treated teeth, providing a realistic model for investigating the initiation, progression, contributing factors, and management of cracked teeth [
19,
29].
This study presents limitations related to specimen selection and laboratory conditions for cyclic load measurements [
11]. Human maxillary central incisors were specifically chosen for this study as they are the type of teeth most susceptible to trauma [
31]. As the use of immature teeth was not feasible, mature teeth were modified to simulate stages of root development, acknowledging the inherent differences in tissue composition [
18].
Histological evaluations of teeth treated with REPs have shown the presence of intracanal ectopic bone, cementum-like tissue, disorganized fibrous connective tissue, and PDL in various combinations [
26,
32]. However, reproducing such biological healing patterns
in vitro is not possible, as natural revascularization cannot be replicated under laboratory conditions. Therefore, root development was mechanically simulated to approximate these clinical scenarios. Protocols for REPs vary widely, particularly regarding the use of scaffolds [
6,
8]. Since blood clot formation is important for tissue regeneration [
8], a foam material was used in this study to simulate a clot under controlled laboratory conditions. Its uniform structure helps standardize the coronal barrier and isolate the mechanical effect of root development. However, foam does not reproduce the complex, viscoelastic, and fibrin-based architecture of a natural blood clot [
8]. Real clots exhibit non-linear, time-dependent behavior and can deform via fibrin stretching or microcrack formation, whereas foam mostly compresses uniformly and collapses under load. Additionally, natural clots can adhere to small dentinal irregularities, whereas foam cannot, a factor that may influence how stresses develop along the canal walls. Finally, blood clots change their mechanical properties over time, while foam remains static, meaning it cannot fully mimic the dynamic nature of clot maturation [
33,
34].
Including a biomaterial control group could potentially enhance external validity; however, the primary objective of this study was to isolate and examine the influence of simulated root development rather than compare materials. Teeth and restorations are naturally exposed to complex oral conditions, including humidity, temperature, pH, and biofilm activity, which contribute to failures [
29,
35]. In this setup, only humidity was controlled using a wet gauze; incorporating additional variables would enhance realism but add complexity and cost.
In vitro PDL simulation remains non-standardized, with variability in materials, application techniques, and layer thickness. No studies have demonstrated a consistent viscoelastic layer capable of producing standardized, clinically relevant tooth mobility during testing [
36]. Heintze
et al. [
37] further questioned the use of artificial periodontium, citing their observation of high variability in silicone layer thickness (300–700 μm), which compromises standardization and leads to uncontrolled tooth mobility under loading. In the present experiment, the use of Erkoskin mitigated these issues. The material can be applied uniformly as a varnish, forming a homogeneous and adherent layer around the tooth root. This root–PDL simulation complex acted as a monoblock, which was then embedded in acrylic resin. This ensured stable fixation without risk of tooth dislocation during testing. This simple and practical approach, however, remains a methodological limitation, as the material’s biomechanical and biological properties require further validation [
36].
The experimental conditions in this study were standardized to match those of previous studies. This included the applied load, the number of loading cycles, test geometries, and instrumentation [
19]. However, fracture resistance testing can be influenced by various factors such as the type of loading device, antagonist material, crosshead speed, tooth embedding technique, and angle of force application [
38,
39]. While masticatory forces vary in direction and intensity, a single 45° oblique force was applied to simulate a typical occlusal function [
11,
24]. Though anatomically shaped metallic antagonists may provide more clinically relevant results, [
39] their high cost and complexity of fabrication make them impractical. In this study, a 2-mm-diameter stainless steel sphere was employed as the loading applicator [
35]. Though the range of normal chewing force has been estimated from 50 to 190 N in the frontal area and 300 to 800 N in the molar area, the initial load used in this study was set at 200 N, representing a subcritical force that exceeds normal masticatory loads but remains below the threshold for catastrophic fracture [
19,
29]. This load level was selected to promote accelerated initiation of microcracks while maintaining a clinically relevant loading condition and an efficient testing duration.
The large standard deviations and high variability observed in this study, with coefficients of variation exceeding 100% in some groups, likely reflect specimen heterogeneity. Despite careful selection, extracted human teeth naturally differ in root morphology, canal anatomy, dentinal thickness, and mineral content. Such variability, along with slight differences in simulated maturation stages, may have affected stress distribution and fatigue behavior during cyclic loading. Additionally, Step-stress fatigue testing is inherently probabilistic, as crack initiation and propagation depend on specimen-specific microscopic flaws. Consequently, wide dispersion in cycles-to-failure and fracture load values is expected. The variability observed thus reflects intrinsic limitations in specimen standardization and may have reduced statistical power to detect subtle intergroup differences. In this study, caliper/radiographic measurements were used to reduce morphological variability; however, the use of micro-computed tomography for full three-dimensional (3D) assessment of canal geometry would provide even more precise matching and is a recommended approach for future studies.
Utilizing 3D-printed standardized samples may further reduce geometric variability and improve reproducibility. Studies should also investigate novel materials to reinforce the cervical area and prevent root fractures. Additionally, incorporating oral environment factors—such as biofilm exposure during cyclic loading—would enhance experimental relevance, as such conditions have been shown to significantly reduce the fatigue resistance of coronal dentine [
40].
CONCLUSIONS
Within the limitations of the present in vitro study, the findings suggest that the stage of root development following REPs does not significantly influence the mechanical reinforcement of immature teeth. Nonetheless, given that this investigation was performed under controlled laboratory conditions, direct extrapolation of these findings to clinical scenarios should be undertaken with caution.
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING/SUPPORT
The authors have no financial relationships relevant to this article to disclose.
-
AUTHOR CONTRIBUTIONS
Conceptualization, Methodology: Gogos C, Kodonas K, Polydora KA. Data curation, Supervision: Gogos C, Kodonas K. Formal analysis, Investigation: all authors. Funding acquisition, Project administration: Gogos C, Kodonas K. Software: Gogos C, Fardi A. Writing-original draft: all authors. Writing-review & editing: all authors. All authors read and approved the final manuscript.
-
DATA SHARING STATEMENT
The datasets are not publicly available but can be obtained from the corresponding author upon reasonable request.
SUPPLEMENTARY MATERIALS
Figure 1.Representative radiographic images of the five study groups. (A) Completely immature teeth immediately after treatment (Group A). (B) Teeth with apical closure (Group B). (C) Teeth with apical closure and wall thickening (Group C). (D) Intact teeth (negative control, Group D). (E) Simulated immature teeth without material placement (positive control, Group E).
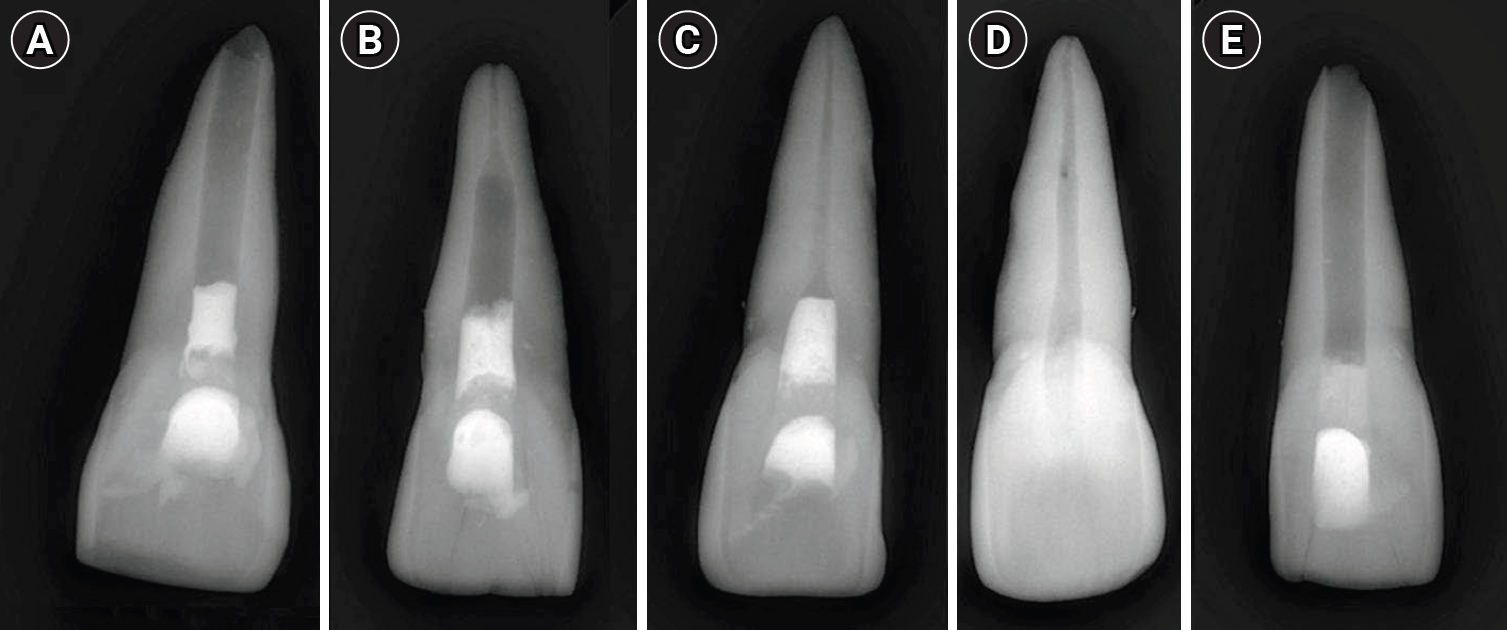
Figure 2.Boxplot representation of mechanical performance across experimental groups. Boxplots illustrate the distribution of (A) number of cycles, (B) cycle number to failure, and (C) load at fracture (N) for the five experimental groups: completely immature, apical closure, apical closure with wall thickening, negative control, and positive control. The boxes represent the interquartile range (IQR), with horizontal lines indicating the median values. Whiskers denote 1.5 × IQR, and outliers are shown as individual points.
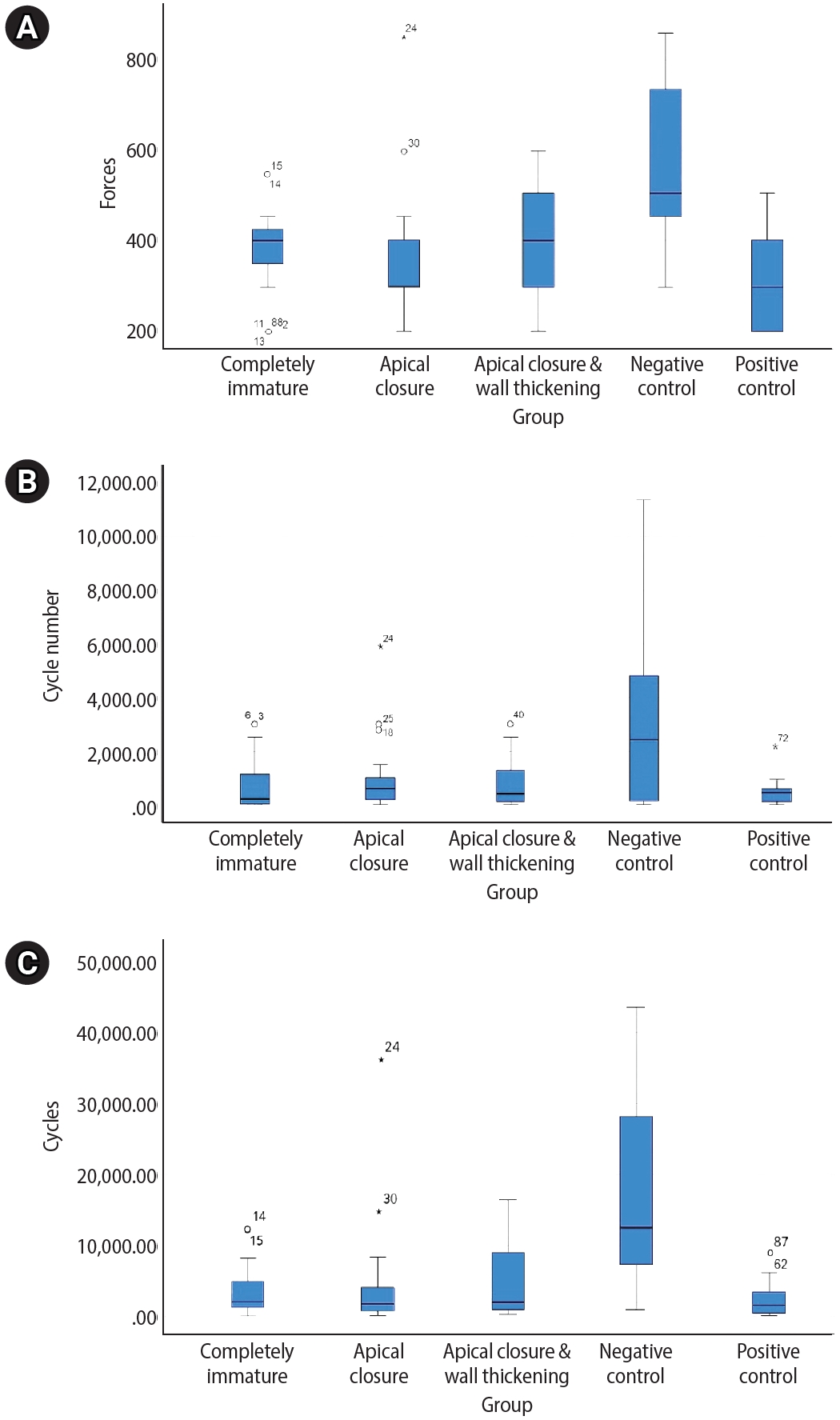
Table 1.Load at failure, fracture resistance, and number of cycles to fracture across groups
|
Group |
Load at failure (N) |
Fracture resistance (MPa) |
Number of cycles |
|
A |
377.50d ± 105.72 (28.00) |
3,686.65d ± 3,300.58 (89.53) |
788.65 ± 1,128.80 (143.13) |
|
B |
362.50d ± 153.79 (42.42) |
4,507.25d ± 7,863.23 (174.46) |
1,009.30 ± 1,438.13 (142.49) |
|
C |
387.50 ± 132.66 (34.23) |
5,019.05 ± 4,968.58 (98.99) |
821.50 ± 893.79 (108.80) |
|
D |
573.33abe ± 186.96 (32.61) |
15,513.33abe ± 13,244.33 (85.37) |
2,972.33 ± 3,334.44 (112.18) |
|
E |
316.67d ± 109.65 (34.63) |
2,513.00d ± 2,797.44 (111.32) |
514.33 ± 549.95 (106.93) |
REFERENCES
- 1. Cvek M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha: a retrospective clinical study. Endod Dent Traumatol 1992;8:45-55.ArticlePubMed
- 2. Cetinkaya A, Ayrancı LB. Evaluation of stress distribution by applied different forces on immature maxillary central teeth with different treatment options: a laboratory finite element stress analysis. BMC Oral Health 2025;25:3.ArticlePubMedPMC
- 3. Andreasen FM, Andreasen JO, Bayer T. Prognosis of root-fractured permanent incisors: prediction of healing modalities. Endod Dent Traumatol 1989;5:11-22.ArticlePubMed
- 4. Araújo PR, Silva LB, Neto AP, Almeida de Arruda JA, Álvares PR, Sobral AP, et al. Pulp revascularization: a literature review. Open Dent J 2017;10:48-56.ArticlePubMedPMCPDF
- 5. Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review: part III: clinical applications, drawbacks, and mechanism of action. J Endod 2010;36:400-413.ArticlePubMed
- 6. Panda P, Mishra L, Govind S, Panda S, Lapinska B. Clinical outcome and comparison of regenerative and apexification intervention in young immature necrotic teeth: a systematic review and meta-analysis. J Clin Med 2022;11:3909.ArticlePubMedPMC
- 7. Huang GT. Apexification: the beginning of its end. Int Endod J 2009;42:855-866.ArticlePubMed
- 8. Wei X, Yang M, Yue L, Huang D, Zhou X, Wang X, et al. Expert consensus on regenerative endodontic procedures. Int J Oral Sci 2022;14:55.ArticlePubMedPMCPDF
- 9. Priya B L, Singh N, Mangalam KK, Sachdev R, P A, Jain HN, et al. Success and complication rates of revascularization procedures for immature necrotic teeth: a systematic review. Cureus 2023;15:e51364.ArticlePubMedPMC
- 10. Nagendrababu V, Murray PE, Ordinola-Zapata R, Peters OA, Rôças IN, Siqueira JF, et al. PRILE 2021 guidelines for reporting laboratory studies in Endodontology: explanation and elaboration. Int Endod J 2021;54:1491-1515.ArticlePubMedPDF
- 11. Mello I, Michaud PL, Butt Z. Fracture resistance of immature teeth submitted to different endodontic procedures and restorative protocols. J Endod 2020;46:1465-1469.ArticlePubMed
- 12. Gunal E, Bezgin T, Ocak M, Bilecenoglu B. Effects of various thicknesses and levels of mineral trioxide aggregate coronal plugs on nanoleakage and fracture resistance in revascularization: an in vitro study. Aust Endod J 2021;47:608-615.ArticlePubMedPDF
- 13. Mello I, Michaud PL, Tanner N. Resistance to fracture of extracted teeth used for pre-clinical endodontic procedures: influence of storage conditions. Eur J Dent Educ 2020;24:272-275.ArticlePubMedPDF
- 14. Cvek M, Andreasen JO, Borum MK. Healing of 208 intra-alveolar root fractures in patients aged 7-17 years. Dent Traumatol 2001;17:53-62.ArticlePubMedPDF
- 15. Jamshidi D, Homayouni H, Moradi Majd N, Shahabi S, Arvin A, Ranjbar Omidi B. Impact and fracture strength of simulated immature teeth treated with mineral trioxide aggregate apical plug and fiber post versus revascularization. J Endod 2018;44:1878-1882.ArticlePubMed
- 16. Elnaghy AM, Elsaka SE. Fracture resistance of simulated immature teeth filled with Biodentine and white mineral trioxide aggregate: an in vitro study. Dent Traumatol 2016;32:116-120.ArticlePubMed
- 17. Galler KM, Krastl G, Simon S, Van Gorp G, Meschi N, Vahedi B, et al. European Society of Endodontology position statement: revitalization procedures. Int Endod J 2016;49:717-723.ArticlePubMed
- 18. Tanalp J, Dikbas I, Malkondu O, Ersev H, Güngör T, Bayırlı G. Comparison of the fracture resistance of simulated immature permanent teeth using various canal filling materials and fiber posts. Dent Traumatol 2012;28:457-464.ArticlePubMed
- 19. Lin F, Ordinola-Zapata R, Xu H, Heo YC, Fok A. Laboratory simulation of longitudinally cracked teeth using the step-stress cyclic loading method. Int Endod J 2021;54:1638-1646.ArticlePubMedPMCPDF
- 20. Li XL, Fan W, Fan B. Dental pulp regeneration strategies: a review of status quo and recent advances. Bioact Mater 2024;38:258-275.ArticlePubMedPMC
- 21. American Association of Endodontists (AAE). Clinical considerations for a regenerative procedure: revised 2021 [Internet]. Chicago, IL: AAE; 2021 [cited 2025 Aug 22]. Available from: https://www.aae.org/specialty/wp-content/uploads/sites/2/2021/08/ClinicalConsiderationsApprovedByREC062921.pdf
- 22. Lenzi R, Trope M. Revitalization procedures in two traumatized incisors with different biological outcomes. J Endod 2012;38:411-414.ArticlePubMed
- 23. Nagy MM, Tawfik HE, Hashem AA, Abu-Seida AM. Regenerative potential of immature permanent teeth with necrotic pulps after different regenerative protocols. J Endod 2014;40:192-198.ArticlePubMed
- 24. Ali MR, Mustafa M, Bårdsen A, Bletsa A. Fracture resistance of simulated immature teeth treated with a regenerative endodontic protocol. Acta Biomater Odontol Scand 2019;5:30-37.ArticlePubMedPMCPDF
- 25. Sharma B, Chalamalasetty N. Modern concepts in endodontic access preparation: a review. Int J Dent Med Sci Res 2024;6:591-594.
- 26. Becerra P, Ricucci D, Loghin S, Gibbs JL, Lin LM. Histologic study of a human immature permanent premolar with chronic apical abscess after revascularization/revitalization. J Endod 2014;40:133-139.ArticlePubMed
- 27. Shimizu E, Ricucci D, Albert J, Alobaid AS, Gibbs JL, Huang GT, et al. Clinical, radiographic, and histological observation of a human immature permanent tooth with chronic apical abscess after revitalization treatment. J Endod 2013;39:1078-1083.ArticlePubMed
- 28. Crozet A, Aubeux D, Pérez F, Gaudin A. Fracture resistance of simulated immature maxillary anterior teeth restored with various canal filling materials, with micro-posts or with a fiber post. Dent Mater J 2023;42:368-374.ArticlePubMed
- 29. Ordinola-Zapata R, Fok AS. Research that matters: debunking the myth of the “fracture resistance” of root filled teeth. Int Endod J 2021;54:297-300.ArticlePubMedPDF
- 30. Jerman E, Lümkemann N, Eichberger M, Hampe R, Stawarczyk B. Impact of varying step-stress protocols on the fatigue behavior of 3Y-TZP, 4Y-TZP and 5Y-TZP ceramic. Dent Mater 2021;37:1073-1082.ArticlePubMed
- 31. Lam R. Epidemiology and outcomes of traumatic dental injuries: a review of the literature. Aust Dent J 2016;61 Suppl 1:4-20.ArticlePubMed
- 32. Martin G, Ricucci D, Gibbs JL, Lin LM. Histological findings of revascularized/revitalized immature permanent molar with apical periodontitis using platelet-rich plasma. J Endod 2013;39:138-144.ArticlePubMed
- 33. Liu D, Nguyen N, Bui TQ, Pocivavsek L. A theoretical framework for multi-physics modeling of poro-visco-hyperelasticity-induced time-dependent fracture of blood clots. J Mech Phys Solids 2025;194:105913.Article
- 34. Mane JV, Chandra S, Sharma S, Ali H, Chavan VM, Manjunath BS, et al. Mechanical property evaluation of polyurethane foam under quasi-static and dynamic strain rates: an experimental study. Procedia Eng 2017;173:726-731.Article
- 35. Lima VP, Machado JB, Zhang Y, Loomans BA, Moraes RR. Laboratory methods to simulate the mechanical degradation of resin composite restorations. Dent Mater 2022;38:214-229.ArticlePubMed
- 36. AlZahrani F, Richards L. Micro-CT evaluation of a novel periodontal ligament simulation technique for dental experimental models. Arch Orofac Sci 2018;13:93-103.
- 37. Heintze SD, Monreal D, Reinhardt M, Eser A, Peschke A, Reinshagen J, et al. Fatigue resistance of all-ceramic fixed partial dentures: fatigue tests and finite element analysis. Dent Mater 2018;34:494-507.ArticlePubMed
- 38. de Abreu RA, Pereira MD, Furtado F, Prado GP, Mestriner W, Ferreira LM. Masticatory efficiency and bite force in individuals with normal occlusion. Arch Oral Biol 2014;59:1065-1074.ArticlePubMed
- 39. Silva GR, Silva NR, Soares PV, Costa AR, Fernandes-Neto AJ, Soares CJ. Influence of different load application devices on fracture resistance of restored premolars. Braz Dent J 2012;23:484-489.ArticlePubMed
- 40. Orrego S, Melo MA, Lee SH, Xu HH, Arola DD. Fatigue of human dentin by cyclic loading and during oral biofilm challenge. J Biomed Mater Res B Appl Biomater 2017;105:1978-1985.ArticlePubMedPDF
 , Konstantinos Kodonas1
, Konstantinos Kodonas1 , Anastasia Fardi2
, Anastasia Fardi2 , Christos Gogos1
, Christos Gogos1




 KACD
KACD
 ePub Link
ePub Link Cite
Cite