Search
- Page Path
- HOME > Search
-
Evaluation of mineral induction ability and cytotoxicity of carbonated hydroxyapatite for pulp tissue regeneration: an
in vitro study - S. Swathi Priyadharshini, Chinnasamy Ragavendran, Anand Sherwood, J. Ramana Ramya, Jogikalmat Krithikadatta
- Restor Dent Endod 2024;49(4):e40. Published online October 29, 2024
- DOI: https://doi.org/10.5395/rde.2024.49.e40
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader ePub
ePub Objectives This study aimed to evaluate carbonated hydroxyapatite (CHA)’s ability for mineral induction and its
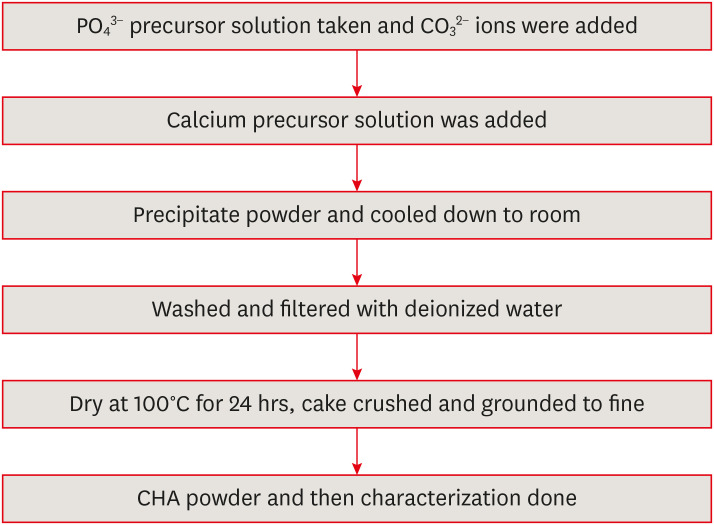
in vitro cytotoxicity with human dental pulp cells.Materials and Methods Precursors for the study include di-ammonium hydrogen phosphate and calcium nitrate tetrahydrate, with sodium hydrogen carbonate added to achieve different levels of carbonate substitution. The synthesized CHA samples are characterized using X-ray diffraction, Fourier transform infrared spectroscopy, and Raman spectroscopy. Scanning electron microscopy (SEM) was used to observe morphology. For 14 days at 37°C, samples were submerged in simulated body fluid to assess their mineral induction capabilities. SEM was used to confirm apatite formation on sample surfaces. The cytotoxicity assay was used to assess the vitality of the cells following their exposure to various concentrations of CHA.
Results The Joint Committee on Powder Diffraction Standards data for HA aligned well with the results from X-ray diffraction analysis of CHA across 3 different concentrations, indicating strong agreement. Fourier transform infrared spectra indicated the presence of phosphate, hydroxyl, and carbonate groups within the samples. SEM and Energy-dispersive X-ray analysis show agglomerated and flaky nanoparticles. All the samples are bioactive, but the formation of apatite differs from one another.
In vitro cytotoxicity assay showed that over 70% of cells maintain viability.Conclusions The results of this study may provide insight into the potential use of carbonated HA as a dental pulp-capping material for vital pulp therapy.
-
Citations
Citations to this article as recorded by- Comparative evaluation of compressive strength and morphological interface of carbonated hydroxyapatite with other pulp capping materials: An in vitro analysis
S. Swathi Priyadharshini, Chinnasamy Ragavendran, I. Anand Sherwood, Ramanaramya Jeyapalan
Endodontology.2025; 37(1): 90. CrossRef - Smart Nanomaterials: Current State and Future Prospects in Drug Delivery and Tissue Engineering
E. Elizabeth Rani, D. Sakthi Sanjana, E. Karthikeyan, J. Nandhini
Biomedical Materials & Devices.2025;[Epub] CrossRef - Thermoresponsive Nanomaterials: Revolutionizing Cancer Theranostics
Bellarmin Michael, Mohanakrishnan Srinivasan, Karthikeyan Elumalai, Lokeshwar Ravikumar, Sivaprakash Kathiresan, Nandhini Jayaprakash
Biomedical Materials & Devices.2025;[Epub] CrossRef - Bioactive Dioxo-Phosphobetaines derived from the reaction of Dichlorodinitrobenzofuroxane with various phosphines
Irina V. Galkina, Haiyan Fan, Semen R. Romanov, Dmitriy I. Bakhtiyarov, Luisa M. Usupova, Svetlana N. Egorova, Yulia V. Bakhtiyarova, Enrico Benassi
Bioorganic Chemistry.2025; 163: 108695. CrossRef - Near-infrared laser-activated PLGA-PDA core-shell nanohybrids for synergistic photothermal antibacterial therapy and sustained ion release in orthodontic white spot lesions prevention
Zezhou Feng, Yujiang Liu, Silu Sun, Minmin Si, Di Huang, Zhiyuan Feng
Journal of Dentistry.2025; 162: 106078. CrossRef - Formation and utilization of soluble microbial products in denitrifying biofilters at different carbon-to-nitrogen ratios: Microbial community characteristics
Fangyuan Jiang, Xianyang Shi
Journal of Environmental Chemical Engineering.2025; 13(6): 119554. CrossRef - Bioactivity and biocompatibility of bioceramic-based pulp capping materials in laboratory and animal models
Rafiqul Islam, Md. Refat Readul Islam, Kenta Tsuchiya, Yu Toida, Hidehiko Sano, Monica Yamauti, Hany Mohamed Aly Ahmed, Atsushi Tomokiyo
Journal of Materials Science: Materials in Medicine.2025;[Epub] CrossRef - Physical, Chemical, and Biological Properties of Graphene Nanoparticle-added Tricalcium Silicate Formulations: A Systematic Review
Soundaria Srinivasan, Deepa Gurunathan, Lakshmi Thangavelu
Journal of International Oral Health.2025;[Epub] CrossRef - Advanced structural and compositional profiling of mineral trioxide aggregate incorporated with nano-carbonated hydroxyapatite: a comprehensive X-ray diffraction and energy dispersive X-ray investigation
Njwan Fadhel Shehab, Nadia Hameed Hasan, Alaa Edrees Dawood, Nawal Atiya Khalaf
Biomaterial Investigations in Dentistry.2025; 12: 216. CrossRef
- Comparative evaluation of compressive strength and morphological interface of carbonated hydroxyapatite with other pulp capping materials: An in vitro analysis
- 2,399 View
- 117 Download
- 8 Web of Science
- 9 Crossref

- Effect of three nanobiomaterials on microhardness of bleached enamel
- Maryam Khoroushi, Farinaz Shirban, Sara Kaveh, Samaneh Doustfateme
- Restor Dent Endod 2016;41(3):196-201. Published online July 14, 2016
- DOI: https://doi.org/10.5395/rde.2016.41.3.196
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader ePub
ePub Objectives The aim of this
in vitro study was to evaluate the effect of incorporating three different nanobiomaterials into bleaching material on microhardness of bleached enamel.Materials and Methods The crowns of 24 extracted sound human molars were sectioned. Sixty enamel specimens (2 × 3 × 4 mm) were selected and divided into five groups (
n = 12): Group 1 received no bleaching procedure (control); Group 2 underwent bleaching with a 40% hydrogen peroxide (HP) gel; Groups 3, 4, and 5 were bleached with a 40% HP gel modified by incorporation of bioactive glass (BAG), amorphous calcium phosphate (ACP) and hydroxyapatite (HA), respectively. The enamel microhardness was evaluated. The differences in Knoop microhardness data of each group were analyzed by one-way ANOVA, followed bypost hoc Tukey tests.Results Significant differences were observed between the study groups. The enamel microhardness changes in Groups 1, 3, 4, and 5 were significantly lower than that of Group 2 (
p < 0.001).Conclusions Within the limitations of this study, it can be concluded that incorporation of each one of the three tested biomaterials as remineralizing agents might be effective in decreasing enamel microhardness changes subsequent to in-office bleaching.
-
Citations
Citations to this article as recorded by- Protective role of calcium-based agents in dental bleaching gels: insights from a systematic review and meta-analysis of clinical and laboratory evidence
Gabriel Pereira Nunes, Renata de Oliveira Alves, Geórgia Rondó Peres, Matheus Henrique Faccioli Ragghianti, Priscila Toninatto Alves de Toledo, Alexandre Henrique dos Reis Prado, Carla Ferreira-Baptista, Alberto Carlos Botazzo Delbem
Clinical Oral Investigations.2025;[Epub] CrossRef - Analysis of Enamel Surface and Shear Bond Strength with Orthodontic Bracket at Different Time Intervals After Bleaching Treatment
Yu-Jin Lee, Ji-Yeon Hong, Hye-Min Ku, Song-Yi Yang
Journal of Dental Hygiene Science.2025; 25(2): 130. CrossRef - Effect of strontium fluorophosphate bioactive glass on color, microhardness and surface roughness of bleached enamel
Shiza Yezdani, Monisha Khatri, Sampath Vidhya, Sekar Mahalaxmi
Technology and Health Care.2024; 32(1): 285. CrossRef - In vitro evaluation of the effect of addition of biomaterials to carbamide peroxide on the bleaching efficacy and microhardness of enamel
Sowmya Kavoor, M. A. Ranjini, Naval Abdul Aziz, H. K. Ashok, Roopa R. Nadig
Journal of Conservative Dentistry and Endodontics.2024; 27(3): 310. CrossRef - Effect of hydrogen peroxide and its combination with nano-hydroxyapatite or nano-bioactive glass on the enamel demineralization and tooth color: An in vitro study
Elham Kheradmand, Alirea Daneshkazemi, Abdolrahim Davari, Maede Kave, Solmaz Ghanbarnejad
Dental Research Journal.2023;[Epub] CrossRef - Over‐the‐counter bleaching agents can help with tooth whitening maintenance
Olívia Santana Jorge, Carolina Noronha Ferraz de Arruda, Rafaella Tonani Torrieri, Rocio Geng Vivanco, Fernanda de Carvalho Panzeri Pires‐de‐Souza
Journal of Esthetic and Restorative Dentistry.2022; 34(2): 328. CrossRef - Effect of Indigenously Developed Nano-Hydroxyapatite Crystals from Chicken Egg Shell on the Surface Hardness of Bleached Human Enamel
Divya Kunam, Vidhya Sampath, Sujatha Manimaran, Mahalaxmi Sekar
Contemporary Clinical Dentistry.2019; 10(3): 489. CrossRef - Influence of Time Intervals between Bleaching Procedures on Enamel Microhardness and Surface Roughness
Roberta Pimentel de Oliveira, Juliana Costa Pereira Baia, Mara Eliane Soares Ribeiro, Mario Honorato da Silva e Souza Junior, Sandro Cordeiro Loretto
The Open Dentistry Journal.2018; 12(1): 555. CrossRef
- Protective role of calcium-based agents in dental bleaching gels: insights from a systematic review and meta-analysis of clinical and laboratory evidence
- 1,503 View
- 7 Download
- 8 Crossref

- Effect of fluoride concentration in pH 4.3 and pH 7.0 supersaturated solutions on the crystal growth of hydroxyapatite
- Haneol Shin, Sung-Ho Park, Jeong-Won Park, Chan-Young Lee
- Restor Dent Endod 2012;37(1):16-23. Published online March 2, 2012
- DOI: https://doi.org/10.5395/rde.2012.37.1.16
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader ePub
ePub Objectives Present study was undertaken to investigate the crystal growth onto synthetic hydroxyapatite (HA) seeds in pH 4.3 and pH 7.0 supersaturated solutions with different fluoride concentrations.
Materials and Methods 8 groups of pH 4.3 and 7.0 calcium phosphate supersaturated solutions were prepared with different fluoride concentrations (0, 1, 2 and 4 ppm). Calcium phosphate precipitates yield crystal growth onto the HA seed surface while solutions flow. For evaluation of crystallizing process, the changes of Ca2+, PO43-, F- concentrations of the inlet and outlet solutions were determined. The recovered solid samples were weighed to assess the amount of minerals precipitated, and finally determined their composition to deduce characteristics of crystals.
Results During the seeded crystal growth, there were significantly more consumption of Ca2+, PO43-, F- in pH 4.3 solutions than pH 7.0 (
p < 0.05). As fluoride concentration increased in pH 4.3 solution, Ca2+, PO43-, F- consumption in experimental solutions, weight increment of HA seed, and fluoride ratio in crystallized samples were increased. There were significant differences among the groups (p < 0.05). But in pH 7.0 solution, these phenomena were not significant. In pH 7.0 solutions, analyses of crystallized samples showed higher Ca/P ratio in higher fluoride concentration. There were significant differences among the groups (p < 0.05). But in pH 4.3 solution, there were not significant differences in Ca/P ratio.Conclusions Crystal growth in pH 4.3 solutions was superior to that in pH 7.0 solutions. In pH 4.3 solutions, crystal growth increased with showed in higher fluoride concentration up to 4 ppm.
-
Citations
Citations to this article as recorded by- Qualitative analysis on crystal growth of synthetic hydroxyapatite influenced by fluoride concentration
Sumi Kang, Jeong Taeg Seo, Sung-Ho Park, Il Young Jung, Chan Young Lee, Jeong-Won Park
Archives of Oral Biology.2019; 104: 52. CrossRef
- Qualitative analysis on crystal growth of synthetic hydroxyapatite influenced by fluoride concentration
- 1,114 View
- 2 Download
- 1 Crossref

- The influence of pH and lactic acid concentration on the formation of artificial root caries in acid buffer solution
- Hyun-Suk Oh, Byoung-Duck Roh, Chan-Young Lee
- J Korean Acad Conserv Dent 2007;32(1):47-60. Published online January 31, 2007
- DOI: https://doi.org/10.5395/JKACD.2007.32.1.047
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader ePub
ePub The purpose of this study is to compare and to evaluate the effect of pH and lactic acid concentration on the progression of artificial root caries lesion using polarizing microscope, and to evaluate the morphological changes of hydroxyapatite crystals of the demineralized area and to investigate the process of demineralization using scanning electron microscope.
Artificial root caries lesion was created by dividing specimens into 3 pH groups (pH 4.3, 5.0, 5.5), and each pH group was divided into 3 lactic acid concentration groups (25 mM, 50 mM, 100 mM). Each group was immersed in acid buffer solution for 5 days and examined. The results were as follows:
1. Under polarized microscope, the depth of lesion was more effected by the lactic acid concentration rather than the pH.
2. Under scanning electron microscope, dissolution of hydroxyapatite crystals were increased as the lactic acid concentration increased and the pH decreased.
3. Demineralized hydroxyapatite crystals showed peripheral dissolution and decreased size and number within cluster of hydroxyapatite crystals and widening of intercluster and intercrystal spaces as the pH decreased and the lactic acid concentration increased.
4. Under scanning electron microscope evaluation of the surface zone, clusters of hydroxyapatite crystals were dissolved, and dissolution and reattachment of crystals on the surface of collagen fibrils were observed as the lactic acid concentration increased.
5. Under scanning electron microscope, demineralization of dentin occurred not only independently but also with remineralization simultaneously.
In conclusion, the study showed that pH and lactic acid concentration influenced the rate of progression of the lesion in artificial root caries. Demineralization process was progressed from the surface of the cluster of hydroxyapatite crystals and the morphology of hydroxyapatite crystals changed from round or elliptical shape into irregular shape as time elapsed.
- 902 View
- 7 Download


 KACD
KACD

 First
First Prev
Prev