Abstract
-
Objectives
Universal adhesives (UAs) are marketed as versatile systems for both self-etch (SE) and total-etch (TE) modes. While their bond strength has been widely investigated, evidence linking fracture characteristics to bonding performance remains limited. This study evaluated the micro-shear bond strength (μSBS) and failure patterns of three UAs applied in SE and TE modes, complemented by fractographic scanning electron microscopy (SEM) analysis.
-
Methods
Eighteen extracted human molars were sectioned to expose mid-coronal dentin and randomly allocated to SE or TE application. Three UAs were tested: Tetric N-Bond Universal, All-Bond Universal, and Single Bond Universal (SBU). Composite micro-rods (n = 72) were bonded, thermocycled for 500 cycles between 5°C and 55°C, and subjected to μSBS testing. Fracture surfaces were examined under SEM and classified as adhesive, cohesive, or mixed. Data were analyzed using two-way analysis of variance, Tukey post hoc test, and Spearman correlation (α = 0.05).
-
Results
In TE mode, SBU demonstrated the highest μSBS (p < 0.001), whereas no significant differences were observed among adhesives in SE mode (p > 0.05). SEM analysis revealed adhesive failures as interfacial fractures, cohesive failures with beach marks, and mixed failures involving crack propagation through both dentin and composite. Adhesive failures correlated negatively with μSBS (rs = –0.77), while mixed failures correlated positively (rs = 0.81).
-
Conclusions
Both the etching strategy and adhesive formulation significantly affect bond strength and fracture behavior. Fractographic SEM analysis provides critical insights into the mechanical reliability of UAs and informs their clinical application.
-
Keywords: Dentin; Dental adhesives; Dental bonding; Fractures, stress; Scanning electron microscopy; Shear strength
INTRODUCTION
The evolution of esthetic dental materials, paired with advancements in adhesive technology, has significantly reshaped restorative dentistry. Modern composites now offer not only lifelike color and translucency but also improved mechanical properties, allowing clinicians to restore both form and function with remarkable precision. Yet even the most visually seamless restoration depends on one critical factor: the strength and reliability of its bond to tooth structure [
1].
Universal adhesives (UAs), classified as eighth-generation bonding agents, were developed to streamline adhesive procedures while maintaining clinical efficacy. Marketed as multimode systems, they promise the strength of traditional total-etch (TE) adhesives with the simplicity and reduced technique sensitivity of self-etch (SE) systems [
2]. These dual capabilities make them a practical choice in daily practice, especially when both speed and performance are desired.
In laboratory settings, the micro-shear bond strength (μSBS) test has emerged as a reliable tool for evaluating dentin–adhesive interfaces. Compared to macro-shear tests, μSBS offers a more consistent stress distribution and fewer internal flaws, thanks to its smaller bonded area [
3,
4]. Moreover, it allows for multiple specimens to be bonded on a single tooth, optimizing the use of biological samples and enhancing data precision [
5,
6]. However, μSBS alone provides only a quantitative assessment of bonding performance and does not reveal the underlying mechanisms of failure. Fractographic analysis, examining the fracture surface post-debonding, has long been a cornerstone in material failure studies. This technique reveals how a material fractured, offering insight into the mode, speed, and origin of failure [
7]. While clinical trials provide real-world performance data, they often fail to isolate the root causes of failure due to the oral environment's complexity. Controlled
in vitro research, on the other hand, can focus on specific variables, making them ideal for analyzing the behavior of adhesives under different bonding conditions [
8].
Despite its potential, the integration of fractographic scanning electron microscopy (SEM) analysis with μSBS testing remains largely unexplored in adhesive dentistry. Our study uniquely combines these complementary approaches to provide both quantitative and qualitative evaluation of UAs, including statistical correlation between bond strength and failure mode. Therefore, this study aims to evaluate the mechanical performance and failure characteristics of three UAs applied in both SE and TE modes, using μSBS testing and fractographic SEM analysis to gain deeper insight into adhesive behavior at the dentin interface. The null hypothesis tested is that there will be no significant difference in bond strength among the UAs when applied in SE or TE modes.
METHODS
Study design
In this
in vitro study, 18 extracted human permanent molars were utilized. Each tooth’s dentin surface was sectioned into four quadrants, with a composite cylinder bonded to each quadrant, yielding a total of 72 composite cylinders for μSBS evaluation. The sample size calculation was based on a power of 80% and α = 0.05, in line with previous findings by El-Safty
et al. [
9]. This method enabled the testing of multiple composite cylinders on a single dentin surface, optimizing the use of extracted teeth [
5]. Teeth were obtained from the Department of Oral Surgery, Faculty of Dentistry, Alexandria University, from patients aged 25 to 45 years and were extracted exclusively for periodontal reasons. Teeth presenting caries, cracks, restorations, attrition, abrasion, or erosion were excluded. Extracted teeth were thoroughly rinsed under running water, stored in a 0.01% (w/v) thymol solution at 4ºC for disinfection [
10], and polished using pumice and rubber cups. Specimens were subsequently stored in isotonic saline at room temperature, with the solution refreshed weekly to ensure hydration and minimize bacterial growth until testing [
10].
Ethical approval was secured prior to the study by the Institutional Ethical Committee, Faculty of Dentistry, Alexandria University (No. 00010556-IORG 0008839). Informed consent agreement was obtained from all patients for undergoing an extraction treatment and for consent to the use of their teeth for research purposes.
Preparation of dentin specimens
Each molar was vertically embedded in a rubber mold (14 mm internal diameter) containing auto-polymerizing acrylic resin, with 2 mm of the root left exposed beneath the cementoenamel junction (CEJ) [
11]. The occlusal surfaces were sectioned with a fine-grit diamond disc under continuous water irrigation to obtain a standardized mid-coronal dentin surface and generate a uniform smear layer, closely simulating clinical conditions [
12,
13]. The cutting disc was replaced after every four teeth to maintain efficiency. All surface preparations were performed by the same operator to minimize variability. After flattening, the specimens were removed from the molds and inspected under a stereomicroscope (Olympus Optical Co. Ltd., Tokyo, Japan) to verify the complete elimination of enamel. The prepared dentin samples were then randomly divided into two experimental groups according to the adhesive approach: SE and TE.
Each main group was divided into three subgroups (
n = 12 specimens per subgroup) according to the UA employed: Tetric N-Bond Universal (TNBU; Ivoclar Vivadent AG, Schaan, Liechtenstein), All-Bond Universal (ABU; Bisco Inc., Schaumburg, IL, USA), and Single Bond Universal (SBU; 3M ESPE, St. Paul, MN, USA). In total, 12 composite micro-rods were bonded per adhesive subgroup (four rods per tooth, using three molars). A summary of the UAs’ characteristics is provided in
Table 1 [
14].
For the TE subgroups, an additional phosphoric acid etching step was carried out before adhesive application, strictly following the manufacturer’s instructions. Composite micro-rods were fabricated with polyethylene tubes (2 mm in height, 0.9 mm inner diameter, 1 mm outer diameter) [
5,
6,
9], which were filled with Filtek Z250 XT composite (3M ESPE). The tubes were placed perpendicularly onto the dentin surface and light-cured for 20 seconds using a light-emitting diode curing unit (Elipar S10; 3M ESPE) positioned directly against the surface to ensure optimal energy delivery, with an output intensity of 1,200 mW/cm² [
15,
16]. After polymerization, the polyethylene molds were gently removed with a No. 11 scalpel blade, leaving four bonded micro-rods per molar. All prepared specimens were stored in distilled water at 37°C for 24 hours before being subjected to thermocycling. Thermal cycling was performed in a custom-built apparatus for 500 cycles between 5°C and 55°C, with a 5-second dwell time at each bath, to simulate intraoral thermal stresses [
17].
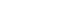
All 72 specimens were subjected to μSBS testing using a universal testing machine (Instron 3345; Instron, Norwood, MA, USA) fitted with a 500 N load cell and operated at a crosshead speed of 0.5 mm/min. A stainless-steel orthodontic wire loop (0.14 mm in diameter) was carefully positioned at the base of each composite rod, in contact with the dentin surface, to apply a shear force parallel to the bonding interface (
Figure 1). The μSBS values were expressed in megapascals (MPa) by dividing the maximum load at failure by the bonded surface area. Failure loads were recorded in newtons (N), and μSBS was determined using the formula ‘σ =
F/A,’ where σ represents μSBS in MPa,
F is the failure load in N, and
A is the bonded surface area in mm
2. The bonded surface area was calculated as
A = π
r2, where π = 3.14 and
r is the radius of each composite cylinder (0.9 mm) [
9].
Following μSBS testing, tooth roots were sectioned at the CEJ using a diamond disc under continuous water irrigation. The obtained dentin slices were gently air-dried to avoid dehydration and minimize the risk of extracting residual monomers or incompletely polymerized oligomers from the fractured surfaces [
17]. Each slice was mounted on a metallic stub and sputter-coated with gold for 1 minute (JFC-1300; JEOL, Tokyo, Japan). The fracture interfaces were then analyzed under a SEM (JSM-IT200, JEOL) operated at 25 kV, with magnifications ranging from ×100 to ×1,000, for detailed characterization and documentation. Failure modes were categorized based on fracture location into four types: adhesive failure (at the adhesive–dentin or adhesive–composite interface), cohesive failure in dentin, cohesive failure in composite, and mixed failure (a combination of adhesive and cohesive). The frequency and percentage distribution of each failure mode were recorded for all groups [
18].
Data were analyzed using Stata/IC software ver. 13.0 (StataCorp LLC, College Station, TX, USA). Mean and standard deviation were calculated for μSBS values. Normality of the data was confirmed using the Shapiro-Wilk test. A two-way analysis of variance (ANOVA) was performed to evaluate the main effects of adhesive system (TNBU, ABU, SBU) and etching mode (SE vs TE) on μSBS, as well as their interaction. Tukey honestly significant difference test was used for pairwise comparisons. Statistical significance was set at p < 0.05. Bar charts were generated to illustrate group comparisons. Spearman correlation was applied to examine the association between μSBS and failure mode.
RESULTS
Micro-shear bond strength assessment
Two-way ANOVA revealed that both the adhesive system and the etching mode significantly affected μSBS (
Table 2). The interaction between the adhesive system and the etching mode was significant (
p = 0.00321). The mean μSBS values for the different adhesives under SE and TE modes are presented in
Table 3. In SE mode, no significant differences were observed among TNBU, ABU, and SBU (
p > 0.05). In TE mode, SBU exhibited significantly higher μSBS compared with TNBU and ABU (
p < 0.001). Within-adhesive comparisons showed that SBU demonstrated a significant increase in μSBS when applied in TE versus SE mode, whereas TNBU and ABU showed no significant difference between the two etching modes.
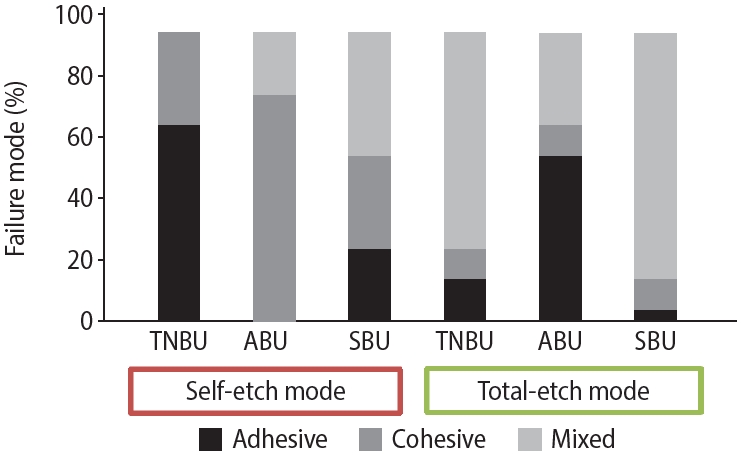
Fractographic evaluation of the fractured specimens showed that adhesive and mixed failures accounted for 45% of the overall failures each. Cohesive failures were observed in 10% of specimens and were limited to four subgroups. In the SE application mode, adhesive failures were more frequent, whereas in the TE mode, mixed failures were more frequent. The percentage distribution of failure modes across all subgroups is shown in
Figure 2.
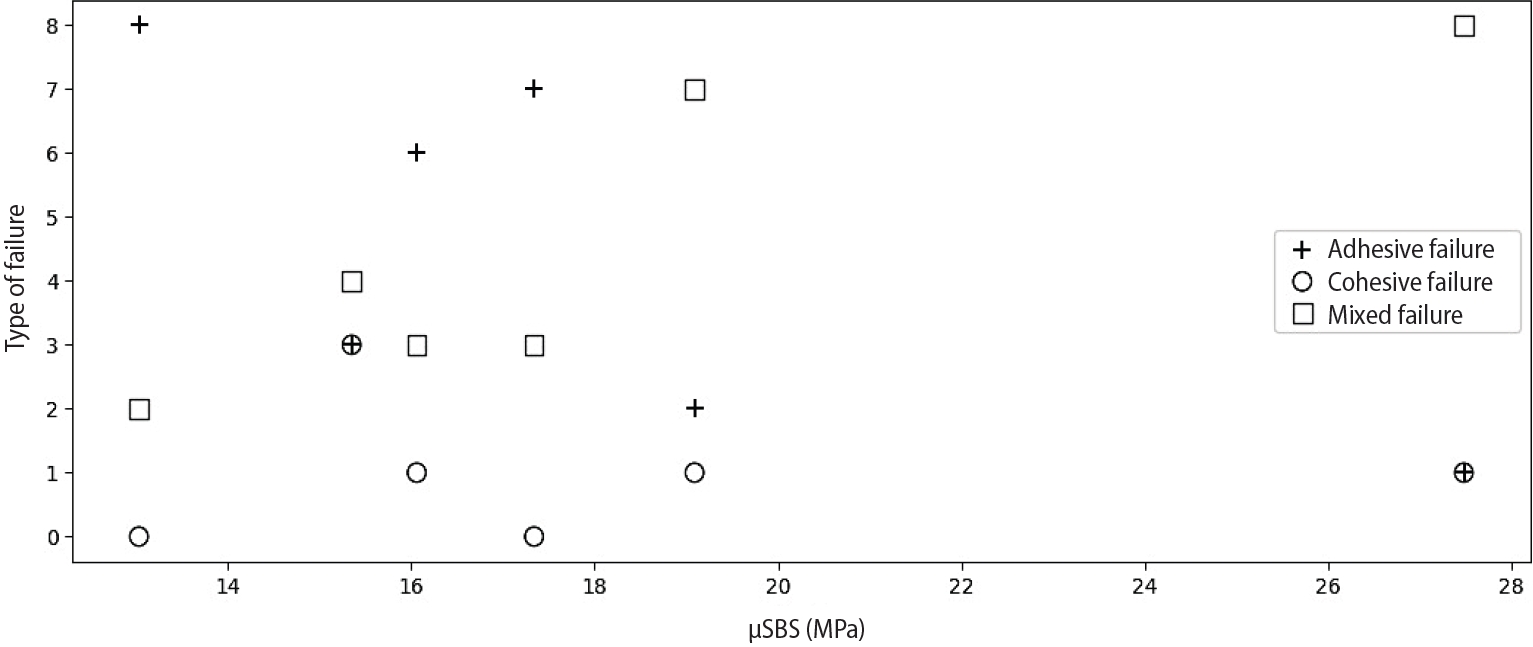
Spearman correlation analysis indicated a negative trend between adhesive failures and bond strength (r
s = –0.77), as well as a positive trend between mixed failures and bond strength (r
s = 0.81). Cohesive failures exhibited a weak positive trend with bond strength (r
s = 0.19). However, none of these associations were statistically significant (
p > 0.05). The ABU subgroup under SE application demonstrated the lowest μSBS (12.83 ± 1.72 MPa) with a predominance of adhesive failures (80%), whereas the SBU subgroup in TE mode exhibited the highest μSBS (26.94 ± 4.22 MPa) with a higher proportion of mixed failures (80%) (
Figure 3).
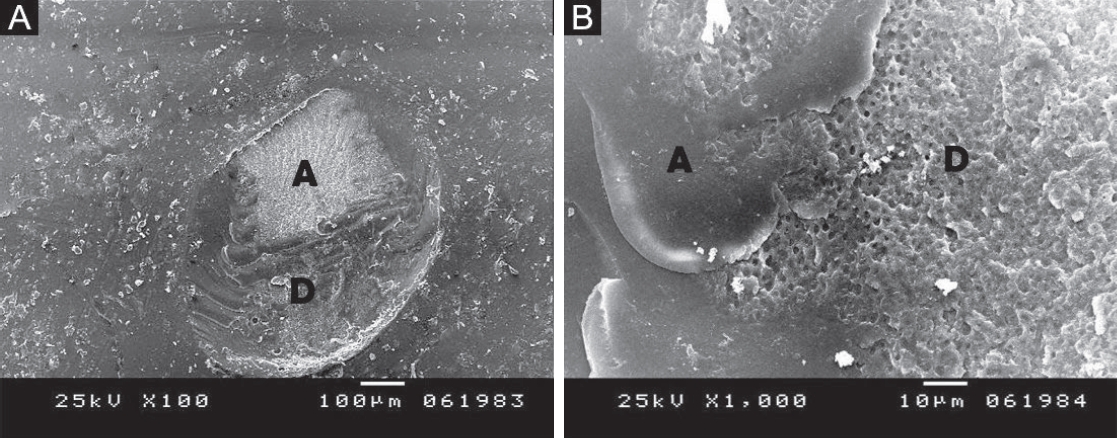
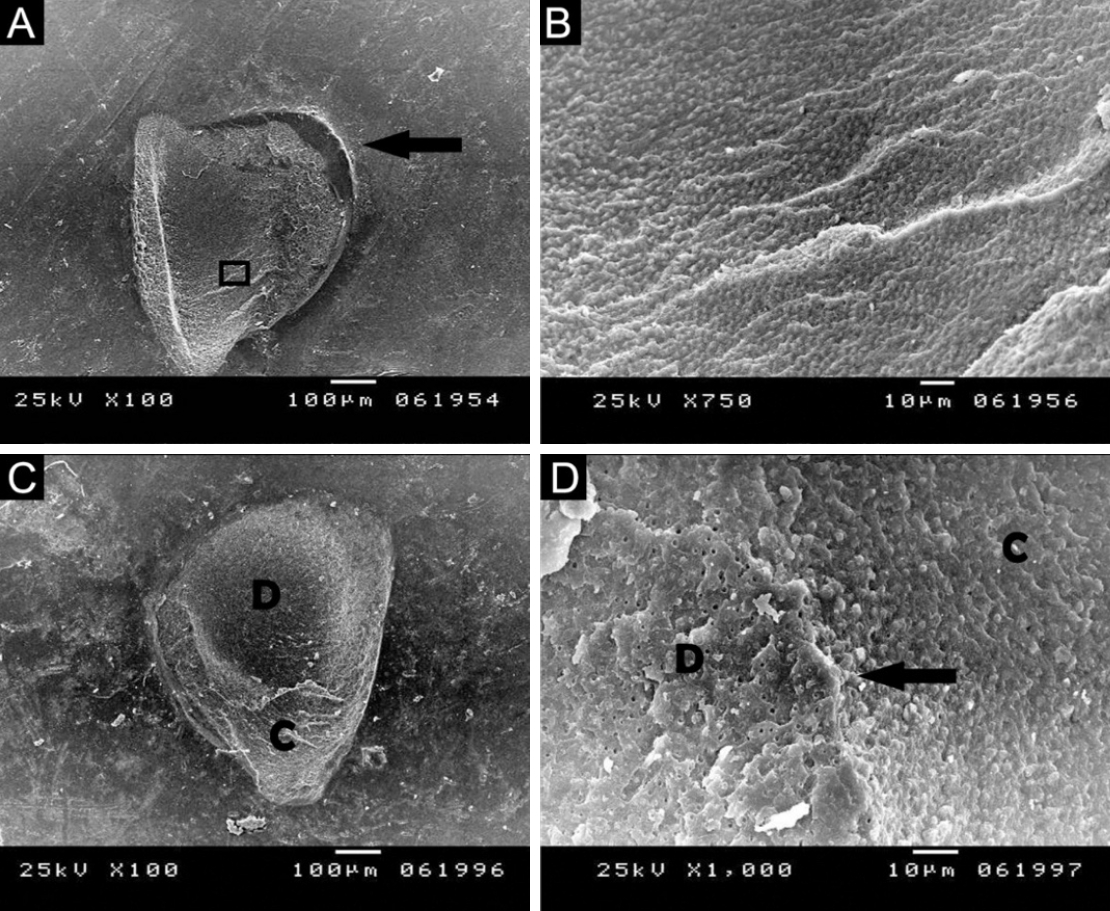
Fractured composite–dentin interfaces were first examined under SEM at low magnification (×100) to locate relevant regions, followed by imaging of selected areas at higher magnifications (up to ×1,000). The analysis identified distinct morphological patterns: adhesive failures were marked by exposed dentin partially covered with residual adhesive; cohesive failures exhibited beach mark features within either dentin or composite; and mixed failures extended across the adhesive layer into both substrates, frequently presenting wavy fracture surfaces and arrest lines, suggestive of progressive crack propagation (
Figures 4–
6).
DISCUSSION
The μSBS test has been advocated as a reliable method for evaluating dentin–adhesive performance, providing more uniform stress distribution and fewer internal defects than macro-shear testing due to its smaller bonded area [
19,
20]. In this study, the null hypothesis was partially rejected. SBU exhibited significantly higher μSBS in TE mode compared to SE mode, whereas TNBU and ABU demonstrated comparable bond strengths across both strategies. This indicates that the influence of phosphoric acid etching depends on adhesive composition. The enhanced performance of SBU under TE conditions can be attributed to its specific formulation, which includes 10-methacryloyloxydecyl dihydrogen phosphate (MDP), a functional monomer that can chemically interact with hydroxyapatite to form stable MDP–Ca salts [
21]. Phosphoric acid etching increases surface energy and exposes collagen fibrils, facilitating greater monomer infiltration [
22,
23]. The presence of bisphenol A-glycidyl methacrylate and 2-hydroxyethyl methacrylate further improves wettability and penetration into the demineralized dentin network [
24]. In contrast, although TNBU and ABU contain functional monomers, differences in solvent type, monomer concentration, and pH appear to reduce their sensitivity to prior acid etching. These findings align with studies reporting that the use of UAs in TE mode does not negatively impact dentin bonding and may even enhance performance in some formulations [
25–
27]. Overall, these results support the versatile application of UAs in both SE and TE modes, while recognizing that certain systems, such as SBU, may benefit more distinctly from phosphoric acid etching.
Failure pattern analysis further supported the μSBS findings. Non-etched dentin predominantly demonstrated adhesive failures, whereas etched specimens exhibited higher proportions of mixed failures, suggesting improved micromechanical interlocking and chemical adhesion after etching. Cohesive failures were uncommon, appearing in only six specimens. SEM confirmed a direct relationship between μSBS and failure mode: mixed failures were associated with higher bond strength, while adhesive failures dominated when μSBS values were lower, consistent with mechanistic observations reported by El-Safty
et al. [
9] and Sabatini [
28]. Overall, the predominance of adhesive and mixed failures over cohesive failures aligns with prior studies by Luque-Martinez
et al. [
29] and Muñoz
et al. [
30] indicating effective resistance to interfacial crack propagation by the tested adhesives. Pre-etching increased mixed failures in SBU, confirming enhanced resin infiltration and stable MDP–Ca bond formation. TNBU and ABU showed less variation with etching, consistent with their comparable μSBS values. Differences from Firat
et al. [
31], who reported more cohesive failures in etched groups, likely reflect variations in adhesive chemistry and methodology. The occurrence of dentin pull-out in some specimens may lead to underestimation of true bond strength in μSBS testing, highlighting the importance of interpreting numerical results alongside fractographic and mechanistic observations [
6].
Fractography serves as an essential tool for relating the stress at failure, the nature of the applied stress, and the dimensions of initial cracks and surrounding microtopography. Characteristic fractographic markings provide insight into failure mechanisms and support more accurate interpretation of adhesive behavior. As a well-established method for failure analysis, fractography is based on the principle that the entire fracture history is recorded on the fractured surface of the tested material [
32]. SEM was employed in this study for fractographic evaluation, revealing the morphological features of dentin and providing insight into failure modes and crack propagation. In cohesive failure micrographs, beach marks (or tide marks) were observed, which represent the incremental positions of a slowly advancing crack, reflecting intermittent or low-level stress within the dentin, as described by Van Meerbeek
et al. [
8]. Other micrographs indicated that when applied stress exceeds the material’s strength, cracks tend to propagate along paths of least resistance, often following dentinal tubules [
33]. In such cases, cracks likely initiated at weak points along the composite–dentin interface and propagated into both dentin and composite, resulting in cohesive failure in both substrates. Analysis of the present findings further suggests that specimens with higher bond strengths exhibited larger fractions of the fractured surface composed of mechanically stronger materials, such as composite resin or dentin. This indicates that cohesive failure occupied a greater area in these specimens, consistent with observations by Hashimoto
et al. [
34].
In this study, the presence of separating slopes was classified as an indicator of mixed failure, reflecting cracks that propagated from the center of the composite microrod toward its periphery at varying depths, shallower on one side, exposing the resin composite, and deeper on the other, exposing dentin. These patterns demonstrate the involvement of both adhesive and cohesive components. In multiple mixed-failure specimens, the adhesive layer acted as the weakest link, with cracks propagating into both the composite and dentin. In some cases, shear forces produced layered separations within the adhesive region, indicating that bond failure was readily initiated in the adhesive zone. In contrast, adhesive-failure micrographs typically exhibited corner-initiated fractures, where failure likely began at the dentin–adhesive interface and progressed through the adhesive layer. Crack nucleation generally occurs at preexisting flaws within materials or at interfaces, such as scratches, voids, or inclusions, which act as stress concentrators under applied loads. The presence of wavy arrest lines in several specimens further reflected localized changes in crack trajectory and transient stress redistribution at the adhesive–dentin interface during rapid μSBS-induced fracture, rather than gradual crack propagation. These observations align with interfacial fracture-mechanics principles and recent reports by Călinoiu
et al. [
7] and Pfeifer
et al. [
35], demonstrating that crack initiation and propagation in bonded interfaces under shear loading are governed by interfacial heterogeneity, local stress intensity, and energy-release patterns. Overall, the observed differences in failure patterns between SE and TE modes suggest that the etching strategy influences local stress distribution and interfacial interactions, consistent with the μSBS outcomes.
Evaluation of bond strength should be complemented by failure mode and fractographic analyses to provide a comprehensive understanding of adhesive resin behavior. Higher μSBS values are generally associated with slower and more stable crack propagation, producing characteristic peaks and valleys indicative of cohesive features. Conversely, lower bond strengths typically correspond to rapid, unstable fractures and a predominance of adhesive failure. These distinctions are consistent with the observations of Hiraishi
et al. [
36], who reported clear morphological differences between specimens exhibiting high and low bond strengths. Moreover, non-uniform stress distribution within the shear zone plays a significant role in shaping failure patterns; complex interfacial stresses can destabilize crack growth and contribute to the occurrence of mixed failures, as documented in earlier investigations [
37–
39].
A key strength of the present study is the combined use of conventional failure mode evaluation and high-resolution fractographic analysis, enabling detailed visualization of crack initiation and propagation paths, providing mechanistic insights into adhesive performance beyond simple bond strength metrics. However, some limitations must be considered. This in vitro study does not fully replicate the dynamic oral environment, where saliva, occlusal loading, biofilm, and pH fluctuations influence adhesive longevity. Only short-term performance after thermocycling was evaluated; no assessment of long-term hydrolytic degradation was performed. Although the μSBS method offers consistent stress distribution, it does not simulate the complex multidirectional forces encountered clinically. The sample size was appropriate for statistical analysis, but it may limit broad generalizability. Finally, only three UAs were examined, and findings cannot be extrapolated to all commercially available formulations. Future studies incorporating long-term durability testing and expanded fractographic evaluation are needed to further elucidate how UAs respond to etching strategies and interfacial stresses.
CONCLUSIONS
Acid etching enhances the bonding performance of UAs, particularly SBU, resulting in predominantly mixed-failure patterns, whereas TNBU and ABU are less affected by etching. The integration of fractographic analysis with conventional failure mode evaluation and bond strength testing provides critical insights into crack propagation and interfacial integrity, offering a comprehensive assessment of adhesive behavior and guiding clinical application strategies.
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING/SUPPORT
The authors have no financial relationships relevant to this article to disclose.
-
AUTHOR CONTRIBUTIONS
Conceptualization, Formal analysis, Methodology: Morsy SM, Holiel AA. Data curation: Morsy SM, Bourgi R, Hardan L, Cuevas-Suárez CE, Holiel AA. Investigation: All authors. Project administration, Supervision, Visualization: Holiel AA. Resources: Morsy SM, Bourgi R, Hardan L, Cuevas-Suárez CE. Software: Morsy SM, Kharouf N, Hardan L, Holiel AA. Validation: Holiel AA, Kharouf N, Bourgi R, Hardan L. Writing - original draft: All authors. Writing - review & editing: Morsy SM, Bourgi R, Holiel AA. All authors read and approved the final manuscript.
-
DATA SHARING STATEMENT
The datasets are not publicly available but are available from the corresponding author upon reasonable request.
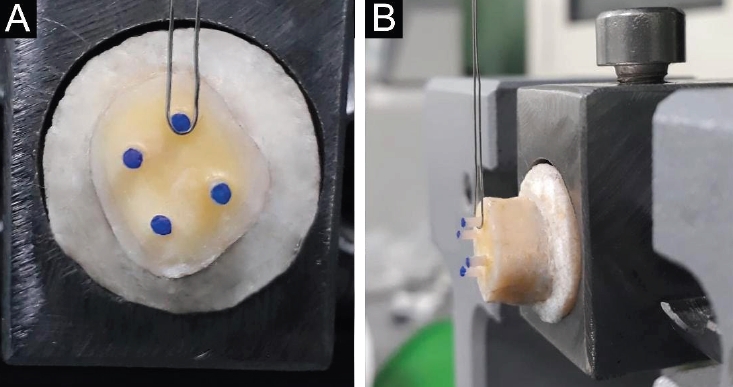
Figure 1.(A) Occlusal view of the resin cylinders. (B) Stainless-steel wire of the universal testing machine positioned perpendicular to the dentin–composite interface.
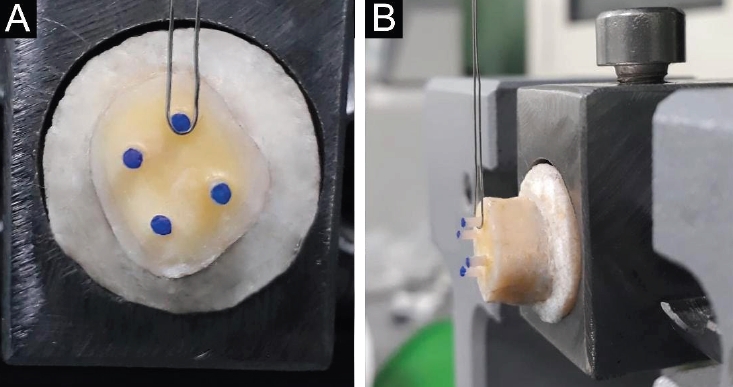
Figure 2.Percentage of failure modes in all study subgroups. TNBU, Tetric N-Bond Universal (Ivoclar Vivadent, Schaan, Liechtenstein); ABU, All-Bond Universal (Bisco Inc., Schaumburg, IL, USA); SBU, Single Bond Universal (3M ESPE, St. Paul, MN, USA).
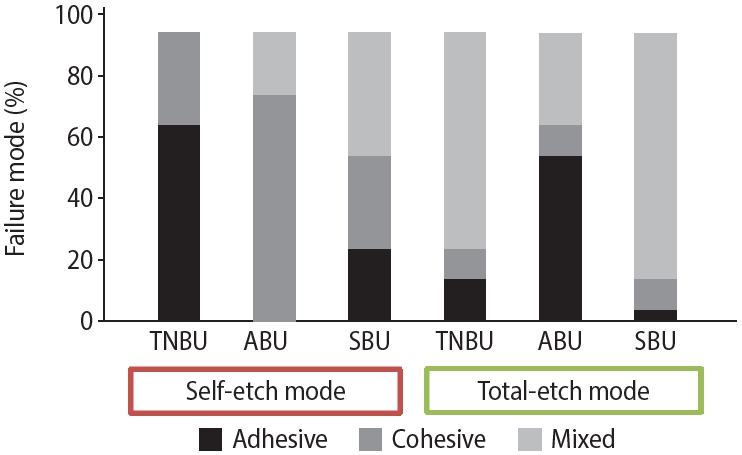
Figure 3.Correlation between micro-shear bond strength (μSBS) values and adhesive, cohesive, and mixed modes of failure.
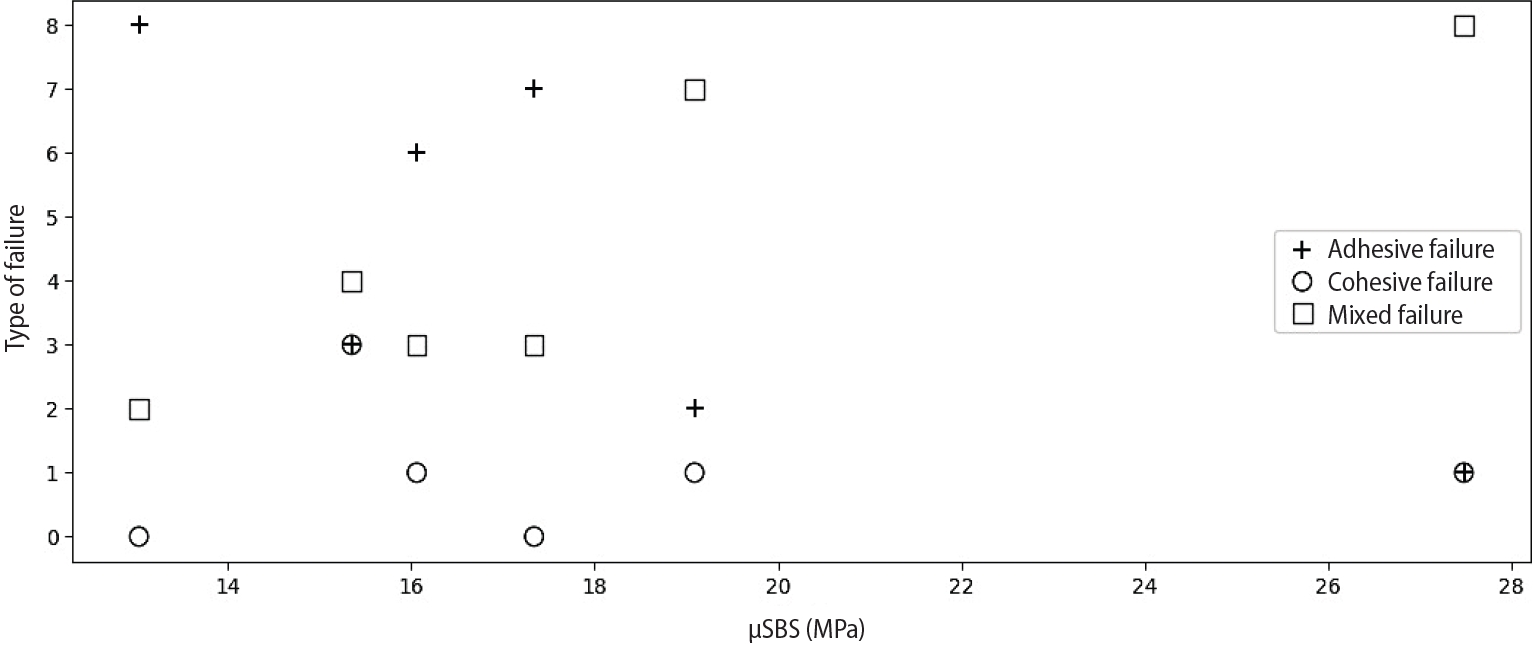
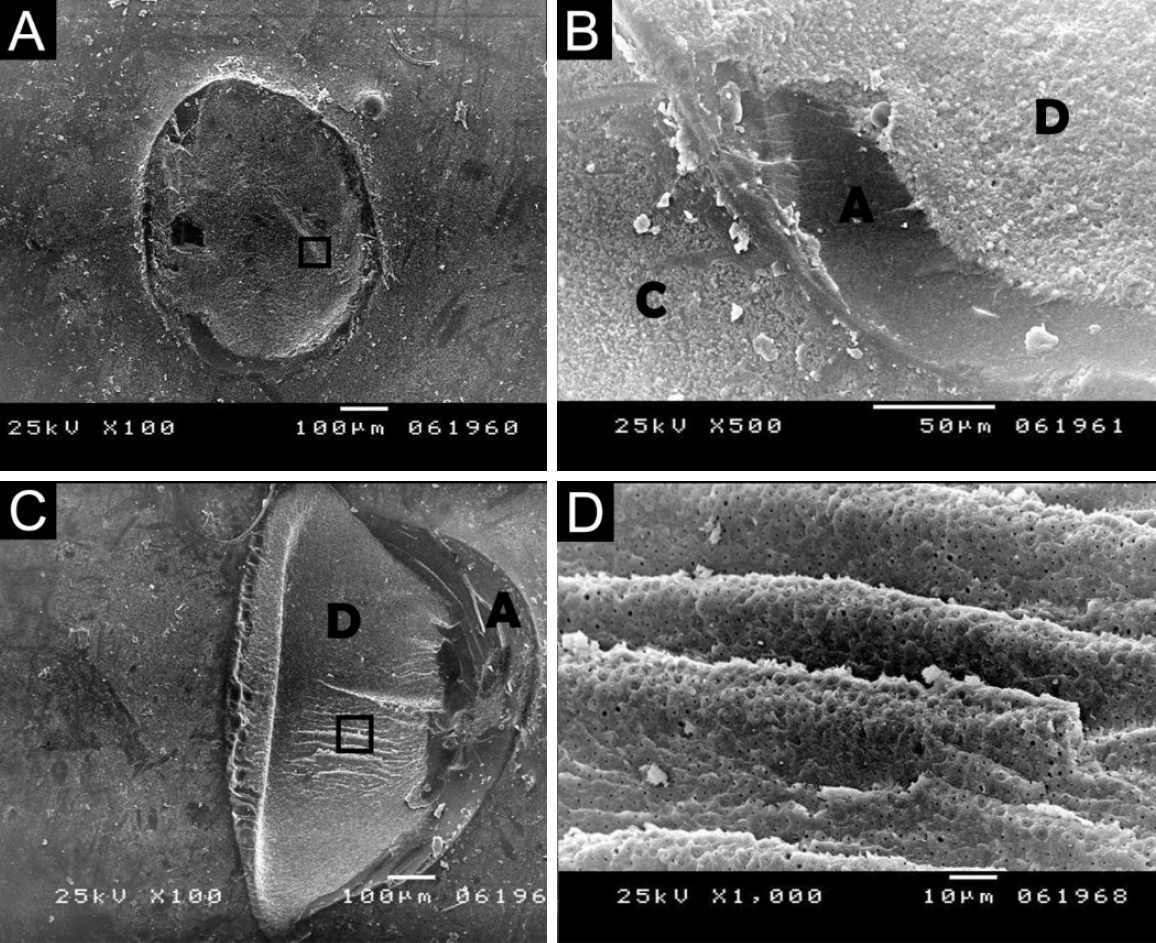
Figure 4.(A) Scanning electron microscopy image showing fracture of the adhesive layer, exposing underlying dentin; failure likely initiated at the dentin–adhesive interface and propagated through the adhesive, indicating adhesive failure. (B) Higher magnification of panel A highlighting adhesive remnants on dentin with no composite traces. (A) ×100; (B) ×1,000. A, adhesive; D, dentin.
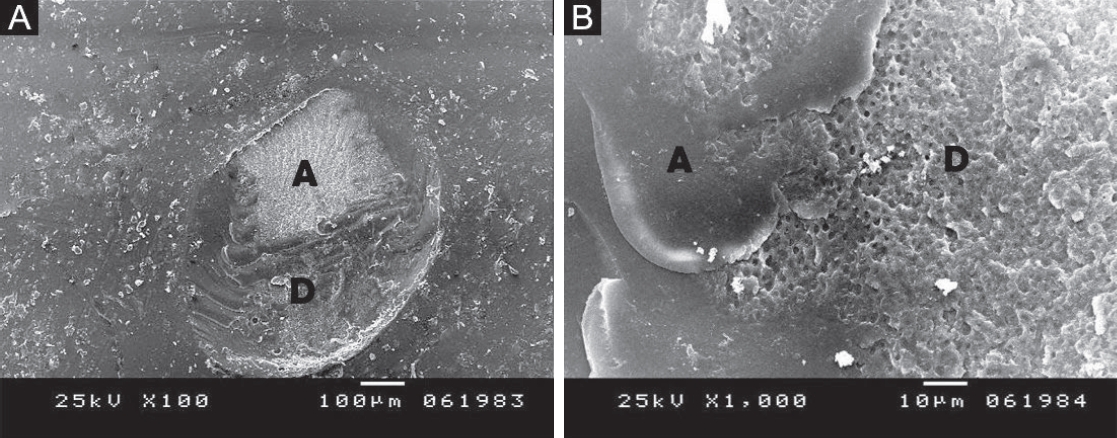
Figure 5.(A) Scanning electron microscopy (SEM) image showing a semicircular flaw (arrow), indicating cohesive failure within dentin. (B) Higher magnification of panel A revealing typical beach marks at the fractured dentin plane, with no composite observed. (C, D) SEM images of the marked area illustrating cohesive failure in both dentin and composite, with the fracture line propagating as a separating slope (arrow). (A, C) ×100; (B, D) ×1,000. D, dentin; C, composite resin.
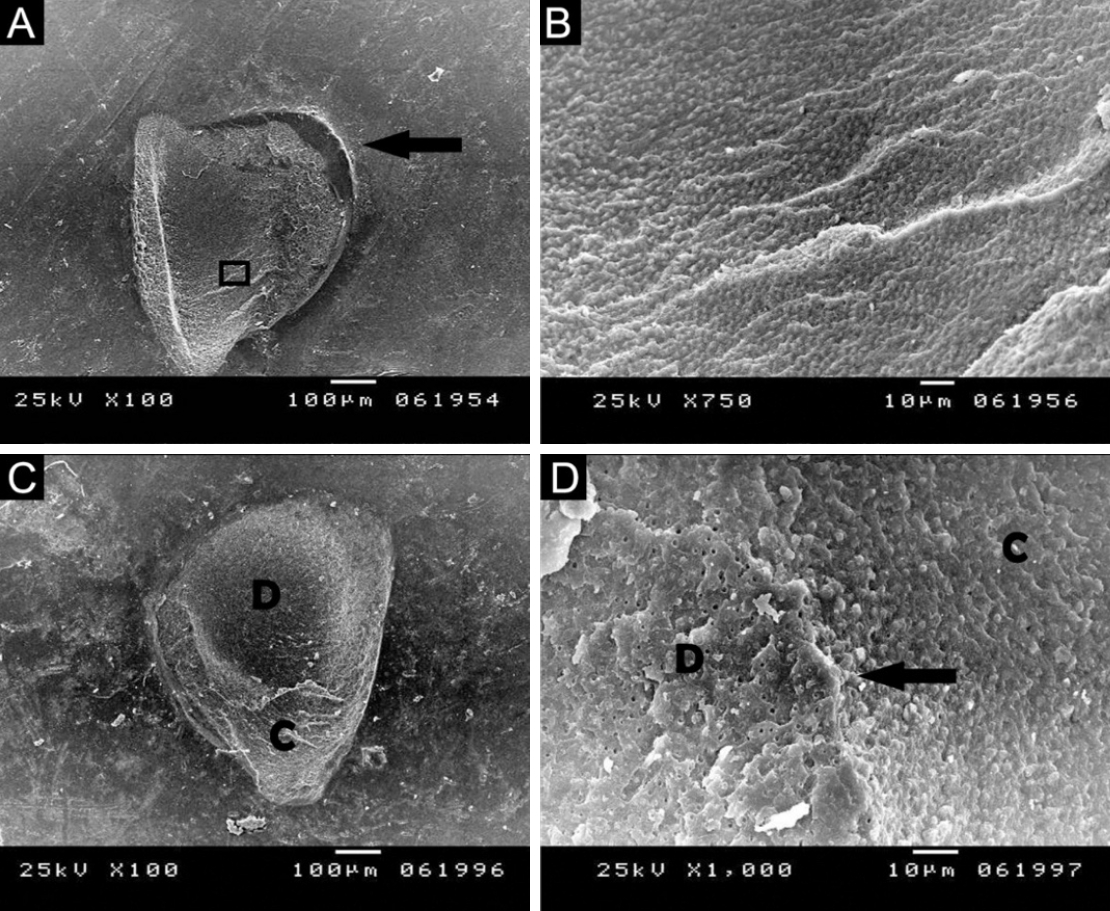
Figure 6.(A, B) Scanning electron microscopy (SEM) images of the marked area showing mixed failure mode extending from the adhesive into dentin and composite, suggesting the adhesive as the crack origin. (C) SEM image also showing mixed failure beginning with the adhesive and propagating into the dentin. (D) SEM of the square-marked area showing a wavy appearance of dentin with arrest lines indicating crack propagation. (A, C) ×100; (B) ×500; (D) ×1,000. A, adhesive layer; D, dentin; C, composite resin.
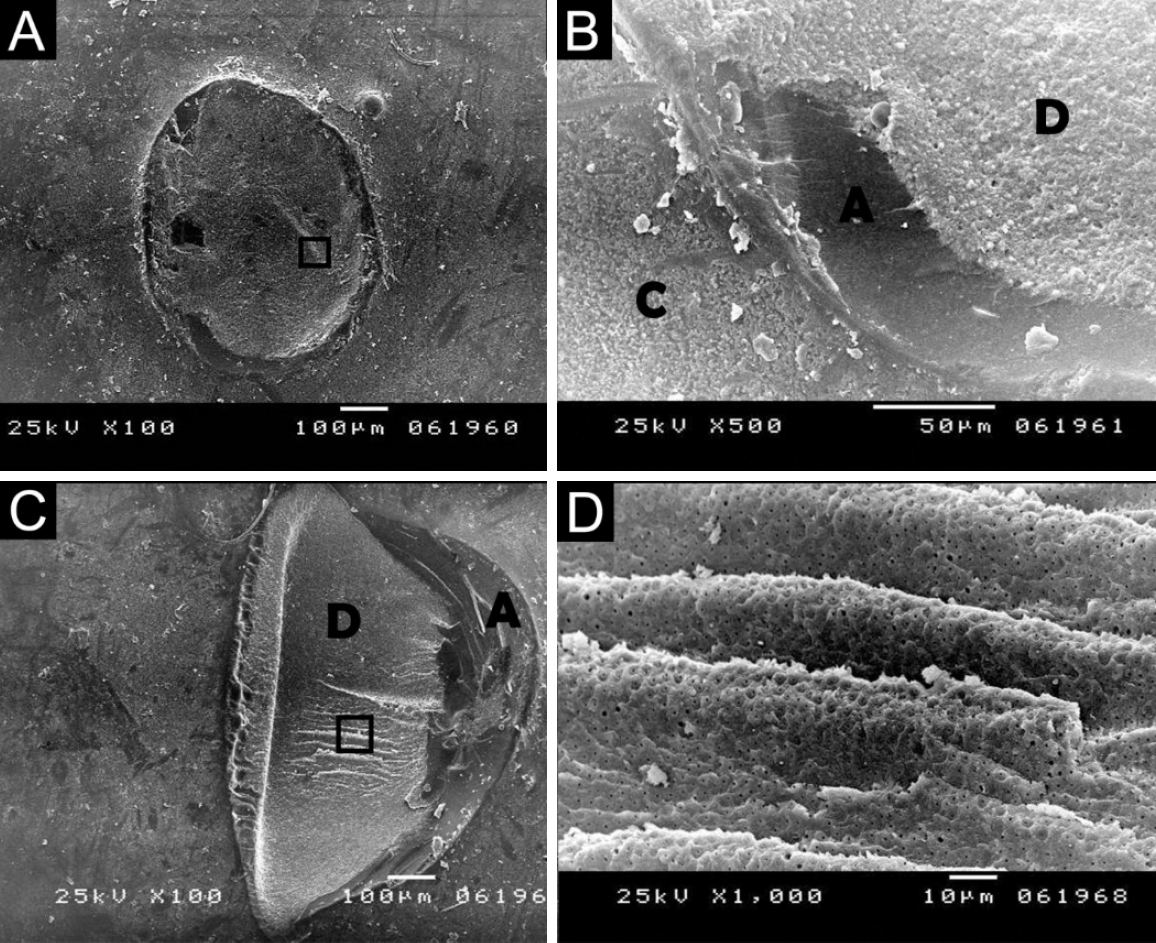
Table 1.Universal adhesives tested in the study, with their respective manufacturers, batch number information, composition, pH, and application mode
|
Adhesive |
Manufacturer (batch No.) |
Composition |
pH |
Self-etch (SE) protocol |
Total-etch protocol |
|
TNBU |
Ivoclar Vivadent, Schaan, Liechtenstein (Z031JT) |
HEMA, Bis-GMA, ethanol, 10-MDP, methacrylated carboxylic acid polymer, camphorquinone, DMAEMA |
2.5–3.0 |
Wash dentin 5 sec, blot dry (moist), apply 1 coat, rub 20 sec, gentle air 5 sec until a stable film forms; light cure 10 sec |
Etch 15 sec, rinse 5 sec, blot dry, apply adhesive as per SE protocol |
|
ABU |
Bisco Inc., Schaumburg, IL, USA (2100005314) |
HEMA, 10-MDP, Bis-GMA, ethanol |
2.5–3.2 |
Wash dentin 5 sec, blot dry (moist), apply 2 coats, rub 20 sec, air dry 5 sec until a stable film forms; light cure 10 sec |
Etch 15 sec, rinse 5 sec, blot dry, apply adhesive as per SE protocol |
|
SBU |
3M ESPE, St. Paul, MN, USA (2216500068) |
HEMA, Bis-GMA, 10-MDP, 2-propenoic acid, ethanol, water, polyalkenoic acid polymer, DMAEMA, filler, camphorquinone, silane |
2.7 |
Wash dentin 5 sec, blot dry (moist), apply 1 coat, rub 20 sec, air dry 5 sec until a stable film forms; light cure 10 sec |
Etch 15 sec, rinse 5 sec, blot dry, apply adhesive as per SE protocol |
Table 2.Two-way analysis of variance for the effects of adhesive system and etching mode on micro-shear bond strength
|
Source of variation |
Degrees of freedom |
Sum of squares |
Mean squares |
F |
p-value |
|
Etching mode (SE vs TE) |
1 |
626.57 |
626.57 |
22.46 |
0.00001*
|
|
Adhesive system |
2 |
504.35 |
252.17 |
9.04 |
0.00033*
|
|
Adhesive × etching interaction |
2 |
349.84 |
174.92 |
6.27 |
0.00321*
|
Table 3.Comparison of all subgroups according to micro-shear bond strength values with pairwise comparisons
|
Adhesive |
No. of samples |
SE |
TE |
|
TNBU |
12 |
16.53 ± 6.14a
|
18.98 ± 7.84a
|
|
ABU |
12 |
12.83 ± 1.72a
|
15.96 ± 3.98a
|
|
SBU |
12 |
14.82 ± 5.61a
|
26.94 ± 4.22b
|
REFERENCES
- 1. El-Salamouny NA, Elmahy WA, Holiel AA. Comparative evaluation of bioactive alkasite-based material in different application modes: a 1-year randomized controlled clinical trial. Odontology 2025;113:1689-1700.ArticlePubMedPMCPDF
- 2. Polesso Patias M, Fernandes-E-Silva P, Carreño NL, Lund RG, Piva E, da Silva AF, et al. Comparative clinical performance of universal adhesives versus etch-and-rinse and self-etch adhesives: a meta-analysis. Clin Oral Investig 2025;29:352.ArticlePubMed
- 3. Hosseini M, Raji Z, Kazemian M. Microshear bond strength of composite to superficial dentin by use of universal adhesives with different pH values in self-etch and etch & rinse modes. Dent Res J (Isfahan) 2023;20:5.ArticlePubMedPMC
- 4. Ahmed B, Hamama HH, Mahmoud SH. Microshear bond strength of bioactive materials to dentin and resin composite. Eur J Dent 2023;17:917-923.ArticlePubMed
- 5. Ismail AM, Bourauel C, ElBanna A, Salah Eldin T. Micro versus macro shear bond strength testing of dentin-composite interface using chisel and wireloop loading techniques. Dent J (Basel) 2021;9:140.ArticlePubMedPMC
- 6. Bahadır HS, Tuğutlu EC. Micro-shear bond strength of an alternative self-etch application of "no wait" universal adhesive to caries-affected and sound dentin in permanent and primary teeth. Oper Dent 2024;49:273-281.ArticlePubMedPDF
- 7. Călinoiu ȘG, Bîcleșanu C, Florescu A, Stoia DI, Dumitru C, Miculescu M. Comparative study on interface fracture of 4th generation 3-steps adhesive and 7th generation universal adhesive. Materials (Basel) 2023;16:5834.ArticlePubMedPMC
- 8. Van Meerbeek B, De Munck J, Yoshida Y, Inoue S, Vargas M, Vijay P, et al. Buonocore memorial lecture: adhesion to enamel and dentin: current status and future challenges. Oper Dent 2003;28:215-235.PubMed
- 9. El-Safty MM, Nour KA, Mustafa DS. Effect of saliva contamination and different decontamination protocols on microshear bond strength of a universal adhesive to dentin. Braz Dent Sci 2024;27:e4159.Article
- 10. Salem-Milani A, Zand V, Asghari-Jafarabadi M, Zakeri-Milani P, Banifatemeh A. The effect of protocol for disinfection of extracted teeth recommended by Center for Disease Control (CDC) on microhardness of enamel and dentin. J Clin Exp Dent 2015;7:e552-e556.ArticlePubMedPMC
- 11. Jayasheel A, Niranjan N, Pamidi H, Suryakanth MB. Comparative evaluation of shear bond strength of universal dental adhesives: an in vitro study. J Clin Exp Dent 2017;9:e892-e896.ArticlePubMedPMC
- 12. Gökce D, Usumez A, Polat ZS, Ayna E. Evaluation of four different adhesive systems' bonding strength between superficial and deep dentin. Materials (Basel) 2025;18:3107.ArticlePubMedPMC
- 13. Gonzaga JA, Ávila AJ, Hardan L, Alonso NV, Suárez CE, Nassar N, et al. Calcium and microhardness quantification in healthy and fluorotic dentin conditioned with a self-etching system: an in vitro study. Dent J (Basel) 2025;13:168.ArticlePubMedPMC
- 14. Arandi NZ. The classification and selection of adhesive agents; an overview for the general dentist. Clin Cosmet Investig Dent 2023;15:165-180.ArticlePubMedPMCPDF
- 15. SG Shirsath, K Bhargava, S Mulay, A Gambhir, R Kakodkar. Comparative evaluation of shear bond strength of TheraBase and composite resins to dentin: an in vitro study. J Stomatol 2025;78:1-5.Article
- 16. Öztürk E, Meric OT, Helvacioglu-Yigit D. Does reducing the acid-etching time increase the micro-tensile bond strength of a bulk fill composite to dentin using different universal adhesives? BMC Oral Health 2025;25:412.ArticlePubMedPMC
- 17. McDonough WG, Antonucci JM, He J, Shimada Y, Chiang MY, Schumacher GE, et al. A microshear test to measure bond strengths of dentin-polymer interfaces. Biomaterials 2002;23:3603-3608.ArticlePubMed
- 18. Elraggal A, Raheem IA, Holiel A, Alhotan A, Alshabib A, Silikas N, et al. Bond strength, microleakage, microgaps, and marginal adaptation of self-adhesive resin composites to tooth substrates with and without preconditioning with universal adhesives. J Adhes Dent 2024;26:53-64.ArticlePubMedPMC
- 19. Eren D, Bektaş ÖÖ, Siso SH. Three different adhesive systems; three different bond strength test methods. Acta Odontol Scand 2013;71:978-983.ArticlePubMed
- 20. Braga RR, Meira JB, Boaro LC, Xavier TA. Adhesion to tooth structure: a critical review of "macro" test methods. Dent Mater 2010;26:e38-e49.ArticlePubMed
- 21. Delgado AH, Ahmed MH, Nunes Ferreira M, Mano Azul A, Polido M, Yoshihara K, et al. Physico-chemical properties and performance of functional monomers used in contemporary dental adhesive technology. J Adhes Dent 2025;27:175-193.ArticlePubMedPMC
- 22. Holiel A, Abdel-Fattah W, El Mallakh B. Bond strength and interfacial morphology of a multimode adhesive resin cement to enamel and dentin. Alex Dent J 2015;40:133-139.Article
- 23. Han F, Sun Z, Xie H, Chen C. Improved bond performances of self-etch adhesives to enamel through increased MDP-Ca salt formation via phosphoric acid pre-etching. Dent Mater 2022;38:133-146.ArticlePubMed
- 24. Alomran WK, Nizami MZ, Xu HH, Sun J. Evolution of dental resin adhesives: a comprehensive review. J Funct Biomater 2025;16:104.ArticlePubMedPMC
- 25. Takamizawa T, Barkmeier WW, Tsujimoto A, Berry TP, Watanabe H, Erickson RL, et al. Influence of different etching modes on bond strength and fatigue strength to dentin using universal adhesive systems. Dent Mater 2016;32:e9-e21.ArticlePubMed
- 26. Wagner A, Wendler M, Petschelt A, Belli R, Lohbauer U. Bonding performance of universal adhesives in different etching modes. J Dent 2014;42:800-807.ArticlePubMed
- 27. Bourgi R, Kharouf N, Cuevas-Suárez N, Lukomska-Szymanska M, Haikel Y, Hardan L. Adhesive systems in dentistry: key components and clinical applications. Appl Sci 2024;14:8111.Article
- 28. Sabatini C. Effect of phosphoric acid etching on the shear bond strength of two self-etch adhesives. J Appl Oral Sci 2013;21:56-62.ArticlePubMedPMC
- 29. Luque-Martinez IV, Perdigão J, Muñoz MA, Sezinando A, Reis A, Loguercio AD. Effects of solvent evaporation time on immediate adhesive properties of universal adhesives to dentin. Dent Mater 2014;30:1126-1135.ArticlePubMed
- 30. Muñoz MA, Luque I, Hass V, Reis A, Loguercio AD, Bombarda NH. Immediate bonding properties of universal adhesives to dentine. J Dent 2013;41:404-411.ArticlePubMed
- 31. Firat E, Gurgan S, Gutknecht N. Microtensile bond strength of an etch-and-rinse adhesive to enamel and dentin after Er:YAG laser pretreatment with different pulse durations. Lasers Med Sci 2012;27:15-21.ArticlePubMedPDF
- 32. Abdelaal AM, Kehela HA, Holiel AA. Fracture resistance and fractographic analysis of pressable glass-ceramics with different partial coverage designs for maxillary premolars. BMC Oral Health 2024;24:1078.ArticlePubMedPMCPDF
- 33. Levrini L, Di Benedetto G, Raspanti M. Dental wear: a scanning electron microscope study. Biomed Res Int 2014;2014:340425.ArticlePubMedPMCPDF
- 34. Hashimoto M, Ohno H, Kaga M, Endo K, Sano H, Oguchi H. In vivo degradation of resin-dentin bonds in humans over 1 to 3 years. J Dent Res 2000;79:1385-1391.ArticlePubMedPDF
- 35. Pfeifer CS, Lucena FS, Tsuzuki FM. Preservation strategies for interfacial integrity in restorative dentistry: a non-comprehensive literature review. J Funct Biomater 2025;16:42.ArticlePubMedPMC
- 36. Hiraishi N, Nishiyama N, Ikemura K, Yau JY, King NM, Tagami J, et al. Water concentration in self-etching primers affects their aggressiveness and bonding efficacy to dentin. J Dent Res 2005;84:653-658.ArticlePubMedPDF
- 37. Abouelleil H, Villat C, Attik N, Grosgogeat B, Farge P. Evaluation of interfacial fracture type using two adhesive systems. Dentistry 2014;4:1000212.Article
- 38. Gönder HY, Mohammadi R, Harmankaya A, Yüksel İB, Fidancıoğlu YD, Karabekiroğlu S. Teeth restored with bulk-fill composites and conventional resin composites; investigation of stress distribution and fracture lifespan on enamel, dentin, and restorative materials via three-dimensional finite element analysis. Polymers (Basel) 2023;15:1637.ArticlePubMedPMC
- 39. Walshaw PR, Tam LE, McComb D. Bond failure at dentin-composite interfaces with 'single-bottle' adhesives. J Dent 2003;31:117-125.ArticlePubMed
 , Rim Bourgi2,3,4,*
, Rim Bourgi2,3,4,* , Louis Hardan3
, Louis Hardan3 , Carlos Enrique Cuevas-Suárez5
, Carlos Enrique Cuevas-Suárez5 , Naji Kharouf4,6
, Naji Kharouf4,6 , Ahmed A. Holiel1,2
, Ahmed A. Holiel1,2








 KACD
KACD
 ePub Link
ePub Link Cite
Cite