Endodontic treatment of a molar-incisor malformation – maxillary first molar: a case report
Article information
Abstract
Molar-incisor malformation (MIM) is a developmental dental anomaly primarily affecting permanent first molars, often accompanied by structural irregularities such as cervical mineralized diaphragms (CMDs) and furcal channels. These anatomical complexities present significant challenges for endodontic treatment. This case report presents the endodontic management of a maxillary first molar diagnosed with MIM—a condition for which root canal treatment is rarely reported. The affected tooth exhibited characteristic features of MIM, including underdeveloped roots, CMD, and an open furcal channel. Initial canal negotiation revealed four buccal canals, but the palatal canal could not be located via conventional access. A separate access approach enabled successful identification, disinfection, and obturation of the palatal canal. Follow-up imaging showed healing of the periapical lesion and favorable clinical outcomes. This case highlights the diagnostic and technical challenges in managing MIM-affected teeth and underscores the importance of advanced imaging, tailored access strategies, and careful material selection to achieve successful endodontic outcomes.
INTRODUCTION
Molar-incisor malformation (MIM), also known as molar-root incisor malformation, is a developmental dental anomaly affecting the permanent first molars, maxillary central incisors, and second primary molars. In most reported cases, the first permanent molars are predominantly involved, exhibiting normal crown morphology but short, thin roots with atypical pulp chamber structures, constricted into straight form. This condition was first reported in 2014 [1,2].
Although the precise etiology of MIM remains unclear, many cases have been associated with significant systemic medical events during early childhood, potentially disrupting normal root development [3,4].
MIM-affected teeth often present with clinical complications, including periodontal bone loss (52.6%), endodontic lesions (50.0%), and combined endodontic-periodontal lesions (28.9%) [5]. Endodontic treatment can be particularly challenging due to structural anomalies such as the cervical mineralized diaphragm (CMD) [2,3,6] and furcal communication [3,7], which may obscure canal pathways or provide alternative routes for bacterial ingress.
The prevalence of MIM remains uncertain; the literature still lacks robust population-level data. However, a recent study conducted in an Australian pediatric dental unit reported a prevalence of 0.47% (5 out of 1,054 children), suggesting that MIM, while rare, may be more common than previously assumed [8].
This report presents the endodontic management of a maxillary first molar diagnosed with MIM, providing new clinical insight into this rare condition.
A CASE REPORT
A 17-year-old male patient presented with a chief complaint of noticing a lesion on the gingiva without experiencing any associated pain (Figure 1A). Clinical examination revealed a sinus tract on the palatal gingiva of the maxillary right first molar (#16) (Figure 1B). The tooth exhibited pain to palpation, while percussion and mobility tests were normal.
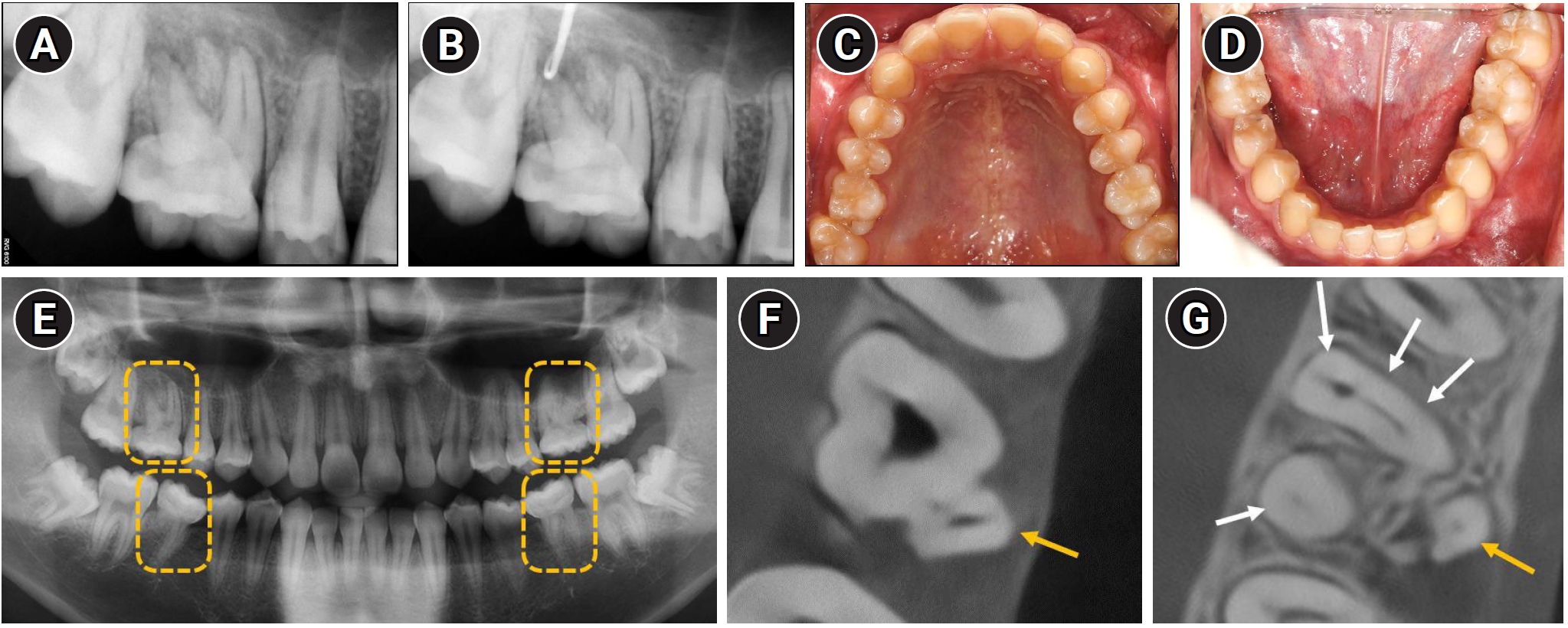
Diagnostic evaluation of maxillary right first molar and overall dentition. (A) Periapical radiograph showing an ill-defined radiolucency associated with tooth #16. (B) Radiograph confirming communication of the sinus tract with the periapical area. (C, D) Intraoral photographs showing normal coronal morphology of all first molars. (E) Panoramic radiograph revealing that all first molars have shortened, tapered roots. Yellow dashed boxes highlight the affected maxillary and mandibular first molars on both sides. (F) Cone-beam computed tomography (CBCT) axial view demonstrating that the palatal canal (yellow arrow) is not in continuity with the pulp chamber. (G) CBCT showing five root canals: three in the mesiobuccal root, and one each in the distobuccal (four white arrows) and palatal roots (yellow arrow).
The patient’s medical history was significant for bilateral arteriovenous malformations diagnosed around the age of one. He underwent endovascular stent implantation under general anesthesia at that time. Since the procedure, he has been undergoing routine follow-up with magnetic resonance imaging every 2 or 3 years. There was no significant dental history related to tooth #16, and the patient had not undergone any prior dental treatment involving the tooth.
Although the coronal morphology of all four first molars was normal (Figure 1C and D), all exhibited root malformations (Figure 1E), leading to a diagnosis of MIM. Based on clinical and radiographic findings, the maxillary right first molar (#16) was diagnosed with pulp necrosis and chronic apical abscess, and nonsurgical root canal treatment was planned.
To obtain a more accurate assessment of the root canal morphology, cone-beam computed tomography (CBCT) was performed. The palatal canal was found to be separated from the main pulp chamber (Figure 1F). Three canals were identified in the mesiobuccal root, and one canal each in the distobuccal and palatal roots, totaling five canals (Figure 1G).
At the first visit, following conventional access cavity preparation, pulp extirpation and working length determination were performed for four buccal canals—three within the mesiobuccal root and one within the distobuccal root (Figure 2A–C). A glide path was established using a HyFlex EDM 10/.05 glide path file (Coltène/Whaledent, Altstätten, Switzerland). Canal shaping was subsequently carried out with a HyFlex EDM 20/.05 taper preparation file, followed by enlargement using a 25/~ HyFlex One File and final finishing using the 40/.04 HyFlex EDM finishing file. Irrigation was performed using 2.5% sodium hypochlorite (NaOCl). For intracanal medication, a calcium hydroxide-based paste (Metapaste Plus; Meta Biomed, Cheongju, Korea) was placed into the canals. The access cavity was temporarily sealed with a temporary restorative material (Caviton; GC Corp., Tokyo, Japan).
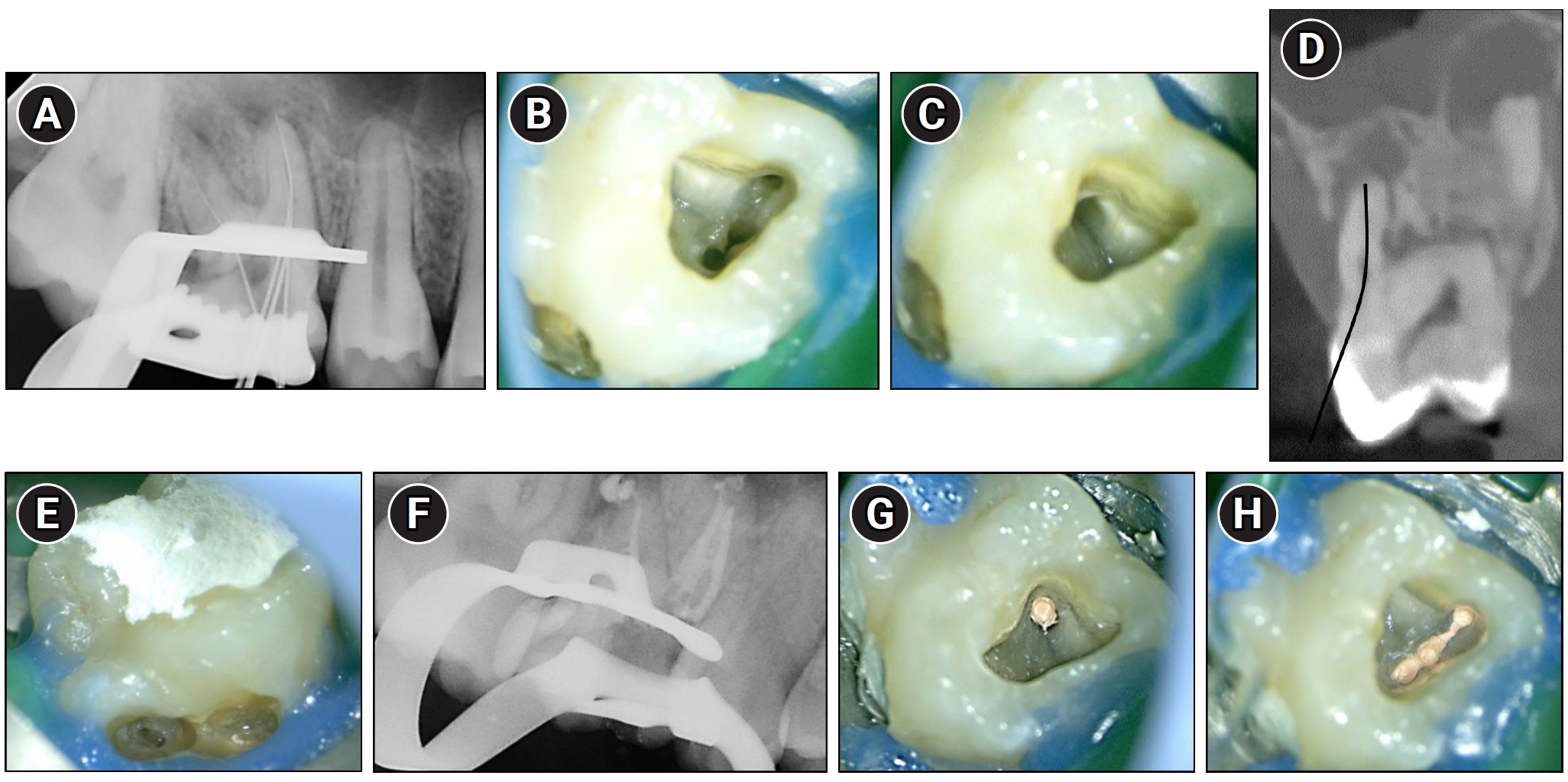
Endodontic management of maxillary right first molar (#16). (A) Working length radiograph taken after conventional access cavity preparation, showing four files in the buccal canals. (B, C) Clinical photographs of the pulp chamber floor showing three canal orifices in the mesiobuccal root and one in the distobuccal root. (D) Cone-beam computed tomography sagittal view showing the cervical access route chosen based on presumed palatal canal location. The black curved line indicates the presumed path for cervical access to the palatal canal. (E) Clinical image showing the perforation area after failed palatal canal identification. (F) Post-obturation radiograph confirming canal filling in three mesiobuccal and one distobuccal canal. (G) Clinical image showing obturation of the distobuccal canal with gutta-percha. (H) Clinical image showing well-condensed gutta-percha filling in the mesiobuccal canals.
As the palatal canal was not connected to the main pulp chamber (Figure 1F), a separate access cavity was planned to locate the palatal canal. An attempt was made to access the canal via a cervical approach, which was deemed appropriate given the canal morphology (Figure 2D). However, the palatal canal could not be identified during the procedure, and a perforation occurred (Figure 2E). The perforation site was subsequently repaired using Biodentine (Septodont, Saint-Maur-des-Fossés, France) and resin-modified glass ionomer (Fuji II LC, GC).
At the second visit, intercommunication among the three mesiobuccal canals was confirmed by observing the flow of 2.5% NaOCl during irrigation. After removing the temporary restoration and intracanal medicament, canal patency was reestablished, and the canals were prepared for obturation. Final irrigation was performed using 2.5% NaOCl, followed by passive ultrasonic irrigation (Endosonic Blue 2; Maruchi, Wonju, Korea). Obturation was performed using a sealer-based technique with a calcium silicate sealer (CeraSeal; Meta Biomed). Following master cone fitting in all four buccal canals, root canal obturation was completed (Figure 2F–H).
At the third visit, to find the palatal canal, a second CBCT scan was obtained. The canal was found to be located in a more mesio-palatal position than initially anticipated (Figure 3A and B). Working length determination, canal instrumentation, and irrigation were subsequently performed using the same protocol and instruments as those employed for the buccal canals.
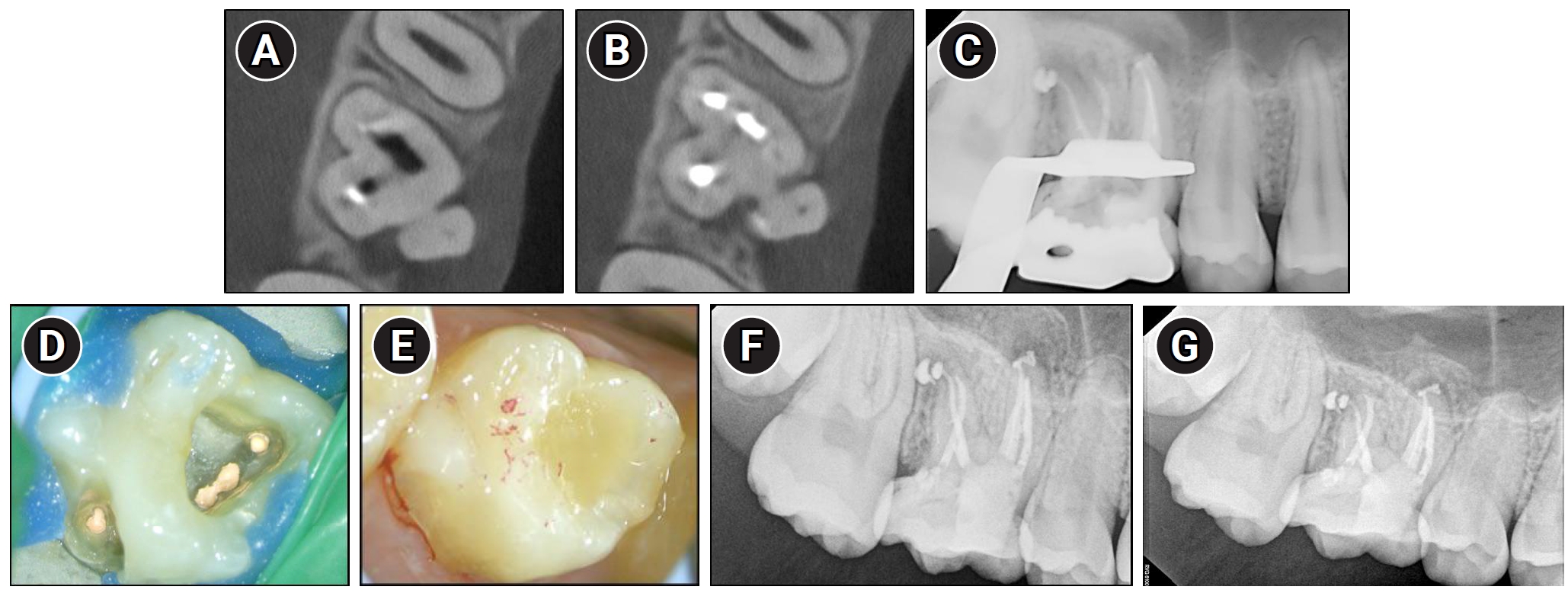
Identification and treatment of the palatal canal through buccal access in maxillary right first molar (#16). (A, B) Re-taken Cone-beam computed tomography axial views showing the palatal canal located more mesially than initially anticipated. (C) Radiograph taken after obturation of the palatal canal. (D) Clinical view of the completed obturation including the palatal canal. (E) Clinical view of the core buildup, demonstrating proper occlusal contacts. (F) One-month follow-up radiographs demonstrating reduction of the periapical radiolucency. (G) Five-month follow-up radiographs showing a further reduction of the periapical radiolucency.
At the following visit, obturation of the palatal canal was performed using the same technique and materials as used for the buccal canals using a sealer-based technique. Core buildup was then completed using bonding (Scotchbond Universal Adhesive; 3M ESPE, St. Paul, MN, USA) and resin core (Filtek Fill and Core, 3M ESPE) (Figure 3C–E).
Serial follow-up evaluations revealed progressive healing, as the periapical lesion showed a gradual reduction in size—initially evident at 1 month (Figure 3F) and continuing to further resolution by 5 months (Figure 3G). The patient is scheduled for continued follow-up evaluations.
DISCUSSION
MIM was reported independently by researchers from Korea and Switzerland in the same year, 2014 [1,2]. Over the past 10 years, since 2014, a total of 24 reports on MIM have been published, with half of them (12 reports) originating from Korean researchers [3]. However, the potential association between MIM and regional or ethnic factors remains unclear and warrants further investigation. Furthermore, although numerous reports have documented the clinical presentation of MIM, only three have described its endodontic management: one involving a maxillary incisor [9] and two involving mandibular first molars [7,10], with no cases reported in maxillary molars.
The first permanent molars are the most commonly affected teeth in MIM. Significant medical events occurring within the first 4 years of life—such as neurological conditions, prematurity, low birth weight, surgical interventions, and medication use—have been frequently associated with this condition. These findings support the hypothesis that early childhood medical history may contribute to the development of this dental anomaly [3,4].
This hypothesis is further supported by the developmental timeline of the first permanent molars. The mineralization of the permanent first molars begins around birth, with crown completion occurring at approximately 3 years of age and root initiation commencing at about 4 years [11]. This period of active dental development overlaps with the time during which many systemic medical events tend to occur, potentially increasing the susceptibility of these teeth to developmental disturbances such as MIM. MIM can manifest with considerable morphological variability, ranging from severely underdeveloped roots to relatively well-formed ones. The present case represents a variant of MIM in which the roots were distinctly separated, highlighting the heterogeneity of this condition and the importance of recognizing its diverse clinical presentations.
One of the common features, and now considered a typical structural anomaly of MIM, is the presence of a CMD—a dense mineralized structure located at the cementoenamel junction [2,3]. Histologically, CMD consists of irregular dentin, osteodentin, or cementum-like tissue, forming a calcified barrier at the cervical region. This structure can obscure the canal pathway or complicate its identification during endodontic treatment. Clinically, CMD hinders access to the root canal system and may lead to misidentification of canal orifices, thereby increasing the risk of iatrogenic errors. Furthermore, numerous microporosities observed within CMD may serve as potential pathways for bacterial ingress and contribute to persistent infection within the pulp chamber, even after treatment [2,3,6].
Another notable feature of MIM is the presence of an open channel in the furcation area. This structure provides direct communication between the external furcation region and the pulp chamber, potentially serving as a pathway for bacterial ingress. Such an anatomical anomaly may increase the risk of persistent infection or reinfection, even after endodontic treatment [3,7]. In addition, the presence of this open communication raises the possibility that immature accessory furcal canals may form at the pulpal floor, further complicating disinfection. These findings highlight the need for careful evaluation of MIM cases when radiolucent lesions persist or recur.
In the present case, both a CMD and an open channel were identified, consistent with previously reported anatomical features of MIM (Figure 4A–C). A periapical lesion developed despite the absence of caries or visible fractures, which are typically associated with pulpal infection. This unusual clinical finding may be explained by the presence of structural anomalies commonly seen in MIM, specifically microporosities within the CMD and an open furcal channel. The CMD-associated microporosities could have acted as microchannels for bacterial ingress, promoting chronic intrapulpal infection. Additionally, the open channel observed in the furcation area may have established a direct connection between the oral environment and the pulp chamber, serving as a potential route for bacterial contamination. Together, these features likely contributed to pulpal pathology and the development of the periapical lesion in the absence of conventional etiological factors.
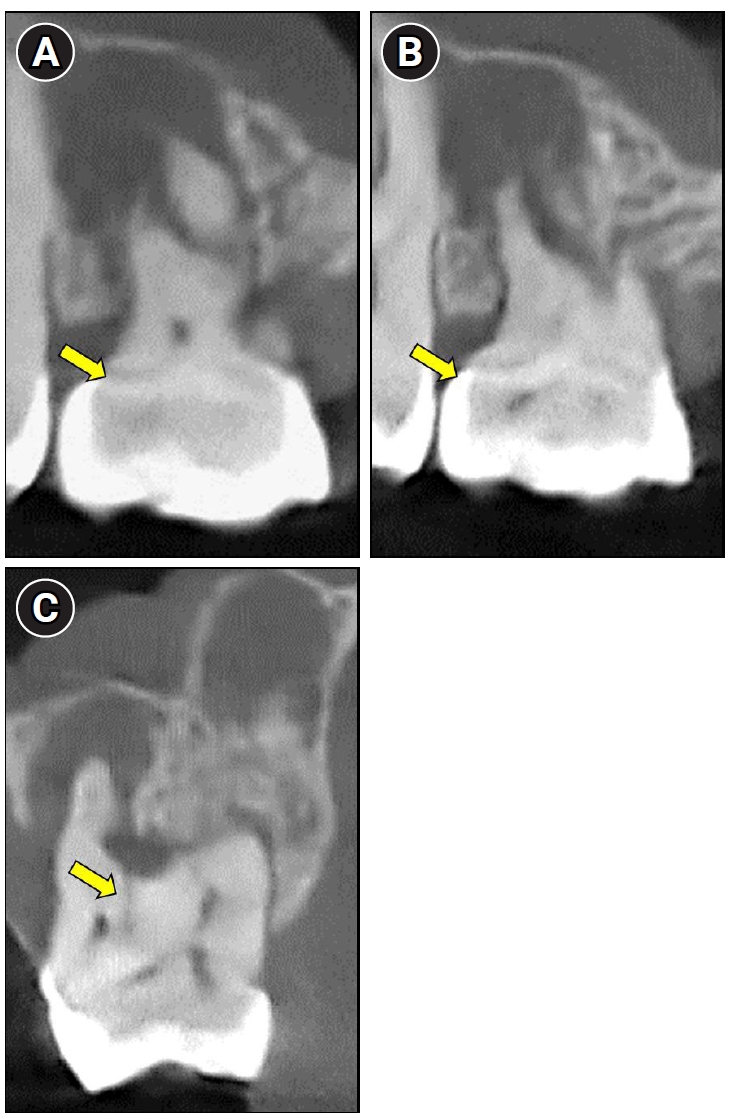
Cone-beam computed tomography images revealing characteristic anatomical features of molar-incisor malformation. (A, B) A dense mineralized structure consistent with a cervical mineralized diaphragm (yellow arrows). (C) An open channel (yellow arrow) connecting the furcation region to the pulp chamber.
In the present case, negotiation of the palatal canal was particularly challenging due to the localized presence of a CMD near the palatal region (Figure 5A and B). The mineralized barrier, with microporosity, likely obscured the true canal orifice, leading to an initial file path error. During the initial attempt, the file may have entered an area presumed to be a microporous structure rather than the true canal pathway, resulting in failure to find the true palatal canal. Although alternative strategies such as guided endodontics or three-dimensional-printed guides could have been considered, these technologies were not available in Korea at the time. We contacted international providers, but confirmed that such services were not accessible locally; thus, they could not be applied in this case.
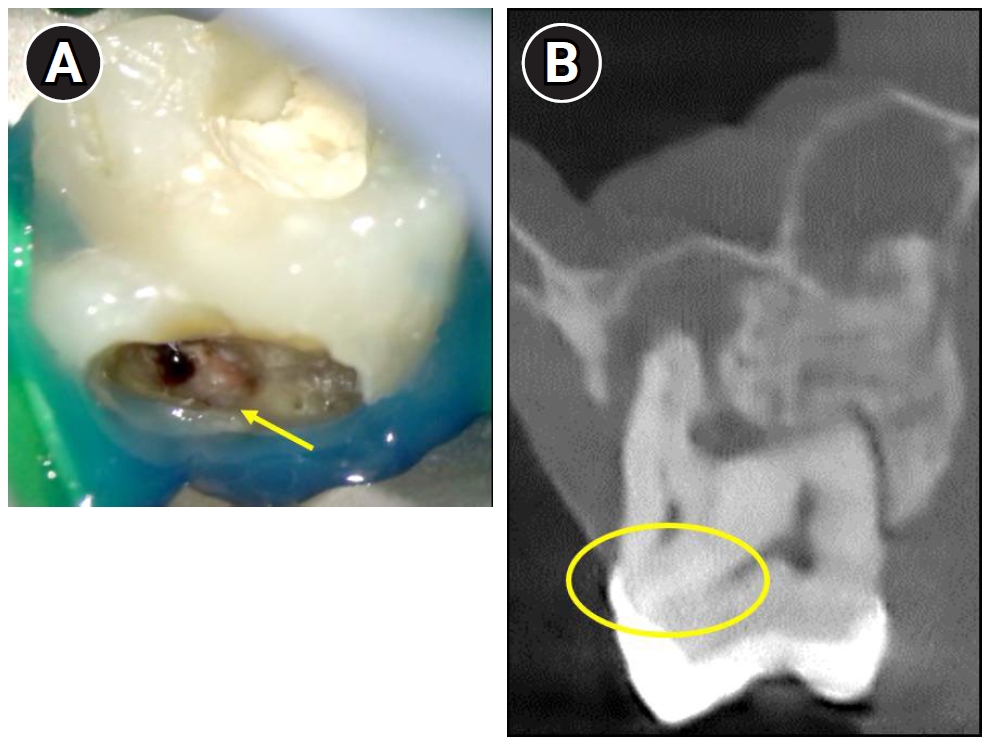
Difficulty in identifying the palatal canal due to the presence of a localized cervical mineralized diaphragm (CMD) near the palatal region. (A) Clinical view showing a mineralized barrier (yellow arrow) obstructing access to the palatal canal. (B) Cone-beam computed tomography image revealing CMD in the palatal area (highlighted with a yellow circle).
While we recognize that continuous wave compaction (CWC) may achieve superior sealing ability compared to sealer-based techniques [12], the clinical circumstances in our case justified a different approach. Specifically, the patient’s young age, the presence of a relatively wide apical foramen at the initiation of root canal treatment, and the need to avoid undue apical pressure all necessitated a more conservative strategy. Therefore, a sealer-based obturation technique with a bioceramic sealer (CeraSeal) was deliberately chosen. Importantly, even if a small amount of sealer extrudes beyond the apex, the favorable biocompatibility of bioceramic sealers minimizes the likelihood of adverse periapical reactions. Moreover, bioceramic sealers exert less apical pressure during obturation, making them particularly advantageous in such cases. This approach not only provided effective sealing in the atypical canal morphology but also leveraged the biological advantages of bioceramic materials in the periapical environment.
Interestingly, the palatal canal was found to be anatomically separated from the main pulp chamber, necessitating access through the palatal cervical region (Figure 2D). Such a configuration, in which the palatal canal is anatomically separated from the main pulp chamber, has not been previously reported in the literature. Therefore, the present case highlights a novel anatomical variation associated with MIM that has not been documented in the existing literature.
Mineral trioxide aggregate (MTA) and Biodentine are widely used for the repair of root perforations due to their biocompatibility and ability to induce hard tissue formation. Compared with MTA, Biodentine has been shown to induce greater RUNX2 (Runt-related transcription factor 2) expression, a key marker of osteoblastic differentiation [13]. It also promotes enhanced hydroxyapatite formation upon contact with tissue fluids, which contributes to improved biological integration and long-term sealing efficacy [13,14]. Clinically, Biodentine offers practical advantages, including a significantly shorter setting time (approximately 12 minutes), higher Vickers microhardness, and superior handling characteristics that improve efficiency and facilitate dentin-like substitution [15,16]. In recent years, premixed MTA putty materials such as EndoSequence Root Repair Material (RRM) Putty (Brasseler USA Dental, Savannah, GA, USA), TotalFill BC RRM Putty (FKG Dentaire Sàrl, Le Crêt-du-Locle, Switzerland), Well-Root PT Putty (VERICOM Co., Ltd., Chuncheon, Korea), and One-Fil PT Putty (MEDICLUS Co., Ltd., Cheongju, Korea) have also been introduced. These newer materials are syringe- or putty-based, user-friendly, set rapidly, and do not induce discoloration. Nevertheless, in the present case, Biodentine was deliberately selected because it is a well-validated material with longstanding clinical validation, offering both favorable handling and biological properties while avoiding tooth discoloration.
CONCLUSIONS
To the best of our knowledge, this is the first documented case reporting endodontic treatment in a maxillary first molar affected by MIM. The tooth exhibited characteristic features of MIM, including a CMD and an open furcal channel, which posed significant challenges to canal identification and disinfection. The palatal canal was found to be separated from the main pulp chamber. Early recognition of CMD, cervical constrictions, abnormal pulp chamber morphology, or root developmental disturbances on radiographs is essential for diagnosing MIM. In treatment planning, obtaining a CBCT scan should be prioritized to accurately assess the complex cervical and furcal anatomy, as CMD and furcal channels can obscure canal pathways or allow bacterial ingress. Careful disinfection and the use of bioceramic sealers and calcium silicate–based materials such as Biodentine are recommended. Long-term follow-up remains critical, and overall, MIM-affected teeth require individualized strategies beyond standard endodontic protocols and, when available, guided endodontic techniques to improve outcomes. This report highlights the anatomical complexity and therapeutic challenges associated with MIM-affected teeth, particularly maxillary molars, and emphasizes the importance of advanced imaging and careful canal-negotiation strategies to achieve favorable clinical outcomes.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING/SUPPORT
The authors have no financial relationships relevant to this article to disclose.
DATA SHARING STATEMENT
The datasets are not publicly available but are available from the corresponding author upon reasonable request.
